Sep. 20, 2011
The National Institutes of Health (NIH) has awarded nearly $2 million to researchers at the Georgia Institute of Technology and Emory University to develop a new class of therapeutics for treating traumatic injuries and degenerative diseases.
The five-year project focuses on developing biomaterials capable of capturing certain molecules from embryonic stem cells and delivering them to wound sites to enhance tissue regeneration in adults. By applying these unique molecules, clinicians may be able to harness the regenerative power of stem cells while avoiding concerns of tumor formation and immune system compatibility associated with most stem cell transplantation approaches.
"Pre-clinical and clinical evidence strongly suggests that the biomolecules produced by stem cells significantly impact tissue regeneration independent of differentiation into functionally competent cells," said Todd McDevitt, director of the Stem Cell Engineering Center at Georgia Tech and an associate professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University. "We want to find out if the signaling molecules responsible for scarless wound healing and functional tissue restoration during early stages of embryological development can be used with adult wounds to produce successful tissue regeneration without scar formation."
In addition to McDevitt, Coulter Department associate professor Johnna Temenoff and Woodruff School of Mechanical Engineering professor Robert Guldberg are also investigators on the project.
Regenerative medicine seeks to restore normal structure and function to tissues compromised by degenerative diseases and traumatic injuries. The contrast between embryonic and adult wound healing suggests that molecules that facilitate tissue regeneration during embryonic development are distinctly different from those of adult tissues.
This grant includes plans for engineering biomaterials that can efficiently capture morphogens, which are molecules secreted by embryonic stem cells undergoing differentiation. The study will also evaluate the regenerative activity of molecule-filled biomaterials in animal models of dermal wound healing, hind limb ischemia and bone fractures. Examining the effects of the morphogens on a range of animal wound models will increase the likelihood of success and define any limitations of the technology, such as its use for specific tissues or injuries.
"Biomaterials have largely been used in an attempt to direct stem cell differentiation or serve as passive cell transplantation vehicles for regenerative medicine and tissue engineering purposes," said McDevitt, who is also a Petit Faculty Fellow in the Institute for Bioengineering and Bioscience at Georgia Tech. "The idea of specifically engineering biomaterial properties to capture and deliver complex assemblies of stem cell-derived morphogens without transplanting the cells themselves represents a novel strategy to translate the potency of stem cells into a viable regenerative medicine therapy."
The award was one of 17 granted this year through the NIH Director's Transformative Research Projects Program (T-R01), which was created to challenge the status quo with innovative ideas that have the potential to advance fields and speed the translation of research into improved health for the American public.
Another T-R01 grant was awarded to Coulter Department professor Shuming Nie, associate professor May Wang and University of Pennsylvania School of Medicine Thoracic Surgery Research Laboratory director Sunil Singhal. That $7 million, five-year grant will support continuing work by the Emory-Georgia Tech Nanotechnology Center for Personalized and Predictive Oncology team on developing fluorescent nanoparticle probes that hone in on cancer cells and on creating instruments that visualize them for cancer detection during surgery.
Since its inception in 2009, the NIH Director's Award Program has funded a total of 406 high-risk research projects, including 79 T-R01 awards.
"The NIH Director's Award programs reinvigorate the biomedical work force by providing unique opportunities to conduct research that is neither incremental nor conventional," said James M. Anderson, director of the Division of Program Coordination, Planning and Strategic Initiatives, who guides the NIH Common Fund's High-Risk Research program. "The awards are intended to catalyze giant leaps forward for any area of biomedical research, allowing investigators to go in entirely new directions."
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Sep. 19, 2011
Genes essential to producing the developmental differences displayed by social insects evolve more rapidly than genes governing other aspects of organismal function, a new study has found.
All species of life are able to develop in different ways by varying the genes they express, ultimately becoming different shapes, sizes, colors and sexes. This plasticity permits organisms to operate successfully in their environments. A new study of the genomes of social insects provides insight into the evolution of the genes involved in this developmental plasticity.
The study, which was conducted by researchers at the Georgia Institute of Technology and the University of Lausanne in Switzerland, showed that genes involved in creating different sexes, life stages and castes of fire ants and honeybees evolved more rapidly than genes not involved in these developmental processes. The researchers also found that these fast-evolving genes exhibited elevated rates of evolution even before they were recruited to produce diverse forms of an organism.
"This was a totally unexpected finding because most theory suggested that genes involved in producing diverse forms of an organism would evolve rapidly specifically because they generated developmental differences," said Michael Goodisman, an associate professor in the School of Biology at Georgia Tech. "Instead, this study suggests that fast-evolving genes are actually predisposed to generating new developmental forms."
The results of the study will be published in the Sept. 20, 2011 issue of the journal Proceedings of the National Academy of Sciences. This research was supported by the National Science Foundation.
The project was an international collaboration between Goodisman, associate professor Soojin Yi and postdoctoral fellow Brendan Hunt from the Georgia Tech School of Biology, and professor Laurent Keller, research scientist DeWayne Shoemaker, and postdoctoral fellows Lino Ometto and Yannick Wurm from the Department of Ecology and Evolution at the University of Lausanne.
Social insects exhibit a sophisticated social structure in which queens reproduce and workers engage in tasks related to brood-rearing and colony defense. By investigating the evolution of genes associated with castes, sexes and developmental stages of the invasive fire ant Solenopsis invicta, the researchers explored how social insects produce such a diversity of form and function from genetically similar individuals.
"Social insects provided the perfect test subjects because they can develop into such dramatically different forms," said Goodisman.
Microarray analyses revealed that many fire ant genes were regulated differently depending on whether the fire ant was male or female, queen or worker, and pupal or adult. These differentially expressed genes exhibited elevated rates of evolution, as predicted. In addition, genes that were differentially expressed in multiple contexts -- castes, sexes or developmental stages -- tended to evolve more rapidly than genes that were differentially expressed in only a single context.
To examine when the genes with elevated rates of evolution began to evolve rapidly, the researchers compared the rate of evolution of genes associated with the production of castes in the fire ant with the same genes in a wasp that does not have a caste system. They found that the genes were rapidly evolving in the genomes of both species, even though only one produced a caste system. These results were also replicated for the honeybee Apis mellifera.
"This is one the most comprehensive studies of the evolution of genes involved in producing developmental differences," Goodisman noted.
This study helps explain the fundamental evolutionary processes that allow organisms to develop different adaptive forms. Future research will include determining what these fast-evolving genes do and how they're involved in the production of different sexes, life stages and castes, said Goodisman.
This project is supported by the National Science Foundation (NSF) (Award No. DEB-0640690). The content is solely the responsibility of the principal investigators and does not necessarily represent the official views of the NSF.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Sep. 06, 2011
Engineers and surgeons are working together to improve the treatment of babies born with craniosynostosis, a condition that causes the bone plates in the skull to fuse too soon. Treating this condition typically requires surgery after birth to remove portions of the fused skull bones, and in some cases the bones grow together again too quickly -- requiring additional surgeries.
Researchers in the Atlanta-based Center for Pediatric Healthcare Technology Innovation are developing imaging techniques designed to predict whether a child's skull bones are likely to grow back together too quickly after surgery. They are also developing technologies that may delay a repeat of the premature fusion process.
"Babies are usually only a few months old during the first operation, which lasts more than three hours and requires a unit of blood and a stay in the intensive care unit, so our goal is to develop technologies that will simplify the initial surgery and limit affected babies to this one operation," said center co-director Joseph Williams, clinical director of craniofacial plastic surgery at Children's Healthcare of Atlanta at Scottish Rite and clinical assistant professor in the Department of Plastic and Reconstructive Surgery at Emory University.
Craniosynostosis affects approximately one in every 2,500 babies in the United States. The condition is caused by the premature closure of sutures with bone. Sutures, which are made of tissue that is more flexible than bone, play an important role in brain growth by providing a method for the skull to increase in size. If the sutures close too soon and get replaced with bony tissue, the skull may limit the normal expansion of the brain.
If untreated, craniosynostosis can cause a range of developmental problems. If treated using the standard treatment course, surgeons remove the fused skull bones, break them up, reposition them, and hold them in place with plates and screws. This usually slows bone growth between the bone pieces, allowing room for expansion of the brain. However, studies show that more than six percent of babies need a second operation to separate the bones again and 25 percent of those require a third operation.
"Following the first surgery, there's a clinical need to be able to screen children on a regular basis to predict when their skull bones are going to fuse together again so that the surgeons can determine if additional intervention will be required," said center director Barbara Boyan, the Price Gilbert, Jr. Chair in Tissue Engineering in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University and associate dean for research and innovation in the Georgia Tech College of Engineering.
To address this need, the researchers have developed a non-invasive technique to monitor bone growth with computed tomography images. They created software that identifies bone in the images, quantifies the distance between the bones, the mass of bone in the gap, and the area and volume of the gap. The research team has demonstrated the utility of this "snake" algorithm using a mouse model of cranial development and recently presented their findings at the 2011 Plastic Surgery Education Foundation conference.
"Using our snake algorithm to analyze computed tomography images of developing skulls in mice, we were able to monitor different types and speeds of bone growth on a daily basis for many weeks," said Chris Hermann, an M.D./Ph.D. student in the Coulter Department. "While one suture fused between 12 and 20 days and then significantly increased in mass for the next 20 days, another came closer together and increased in mass but remained largely open."
The research team recently adapted the technology for use in children and began a clinical study to determine the effectiveness of the algorithm to diagnose cases of craniosynostosis. The researchers hope this technology will improve the ability of physicians to diagnose and determine the severity of craniosynostosis.
In addition, the researchers are studying the biological basis of the condition and developing technologies they hope will delay bone growth and eliminate the need for additional operations. In one project, Coulter Department research scientist Rene Olivares-Navarrete and Williams are examining individuals with craniosynostosis to identify genes that influence suture fusion. Determining the genes that control suture closure may help the researchers identify potential therapeutic targets to prevent premature suture fusion.
The research team has also designed a gel to be injected into the gap created between skull bones during the first surgery. The material -- called a hydrogel because it contains a significant amount of water -- would deliver specific proteins to the area to delay, but not prevent, bone growth.
"The hydrogel cross-links spontaneously because of a reaction between a polyethylene-glycol monomer and a cross-linking molecule, allowing for polymerization without the use of chemical initiators or the production of free radicals," explained Hermann.
Preliminary results in a mouse model of cranial development indicate that the gel, developed in collaboration with Coulter Department associate professor Niren Murthy, can be injected into a gap between skull bones, firm up rapidly and not injure underlying soft tissues or impair bone healing. These pre-clinical results were presented at the Society for Biomaterials Annual Meeting in April 2011.
Both Boyan and Williams see promise in using these technologies to improve the treatment of children with craniosynostosis and eliminate additional operations sometimes needed to treat the condition.
"During the initial surgery, injecting the gel may reduce the operation's severity if it eliminates the need for plates and screws to hold the skull bones in place afterward," explained Boyan, who is also a Georgia Research Alliance (GRA) Eminent Scholar. "After the surgery, if the computed tomography images tell us that the skull is closing too quickly, we may be able to inject the gel through the skin overlying the skull without surgery to further delay the bones from fusing."
The researchers are currently improving the protein release kinetics of the hydrogel and conducting pre-clinical experiments to determine which proteins successfully delay bone growth when included in the gel. Approval from the Food and Drug Administration will be required before this system and hydrogel can be used as a treatment for craniosynostosis.
The Center for Pediatric Healthcare Technology Innovation is supported by Children's Healthcare of Atlanta, in collaboration with Georgia Tech.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Aug. 09, 2011
A new study into the causes of leakage in one of the heart's most complex valve structures could lead to improved diagnosis and treatment of the condition.
An estimated 1.6 million Americans suffer moderate to severe leakage through their tricuspid valve, a complex structure that closes off the heart's right ventricle from the right atrium. Most people have at least some leakage in the valve, but what causes the problem is not well understood.
A new study, published online in the journal Circulation on August 1, 2011, found that either dilating the tricuspid valve opening or displacing the papillary muscles that control its operation can cause the valve to leak. A combination of the two actions can increase the severity of the leakage, which is called tricuspid regurgitation.
"We think this is the first in vitro investigation into the mechanics of the tricuspid valve, and that our findings into the mechanisms that cause tricuspid regurgitation could lead to improved diagnosis and treatment," said Ajit Yoganathan, Regents professor and Wallace H. Coulter Distinguished Faculty Chair in Biomedical Engineering in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
The tricuspid valve consists of three flaps that open to allow blood to flow from the heart's upper right chamber to the ventricle. To close the valve, the flaps re-cover the opening, keeping blood from flowing back into the chamber it just left. When the valve is leaky or doesn't close tightly enough, blood flows backward into the chamber just after the heart contracts.
Tricuspid regurgitation has been increasingly recognized as a source of disease in patients with chronic mitral valve regurgitation, but surgical repair of the tricuspid valve alone is recommended only in rare cases. If an individual suffers from severe tricuspid regurgitation, surgeons will sometimes repair the tricuspid valve during a surgery to repair other leaky heart valves.
"Standard clinical procedures that detail when and how tricuspid valve repairs should be performed need to be developed and this study suggests several items that should be considered," said one of the study’s co-authors David H. Adams, chair of the Department of Cardiothoracic Surgery at Mount Sinai Medical Center in New York City. "Current repairs for tricuspid regurgitation focus mainly on remodeling the valve's opening to correct enlargement, but this study shows that it may also be important to restore the position of the papillary muscles, providing as much overlap as possible, in order to conduct effective and durable tricuspid valve repairs."
With funding from the American Heart Association, Yoganathan and Coulter Department graduate student Erin Spinner conducted experiments with porcine tricuspid valves to determine possible causes of tricuspid regurgitation. The valves were attached to a plate designed to create physiological dilation and then placed inside a right heart simulator.
The researchers first investigated the effect of dilating the opening of the tricuspid valves. When the openings stretched to an area 40 percent larger than their normal size, a hole appeared in the central region of the valve. The hole caused leakage, and increased in size with further dilatation. This finding surprised the researchers because similar studies using the same method had shown that the heart's mitral valve could withstand dilation of 75 percent before leaking.
"These results tell us that the tricuspid valve is a much more complex valve than the mitral valve, which only has two flaps," explained Spinner. "Forming a proper seal over the valve opening might be more difficult with three flaps, which could be why such a large percentage of the population experiences some level of tricuspid regurgitation and why some individuals with annular dilation have tricuspid regurgitation and others do not."
The research team also investigated the effect of displacing the valve's three papillary muscles, which attach to the valve via threads. Contraction of the papillary muscles opens the valve and relaxation of the muscles closes the valve. The study showed that papillary displacement alone resulted in significant tricuspid leakage.
"While isolated displacement of the papillary muscles is rare, these results are relevant toward understanding what may happen if the size of the valve opening is repaired, but the papillary muscles are left displaced," noted Yoganathan.
The study also showed that higher levels of tricuspid leakage resulted when the researchers combined the conditions -- dilation of 40 percent or greater and displacement of all papillary muscles.
In their future work, the Coulter Department researchers plan to look at the effect of pulmonary hypertension on the tricuspid valve, because tricuspid regurgitation usually develops in association with pulmonary hypertension -- which is abnormally high blood pressure in the lungs. They also plan to work with their clinical collaborators to extend their findings to humans.
"In our in vitro study we were able to select specific porcine valves, but with human subjects there will be more anatomical variety. For example, two people may have valves of the same diameter, but one person may have longer flaps that are able to compensate for dilation whereas the other might not," noted Yoganathan.
In addition to those already mentioned, Pedro del Nido, a professor in the Department of Cardiothoracic Surgery at Children's Hospital Boston; Emir Veledar, an assistant professor with a joint appointment in the Division of Cardiology and Division of Epidemiology at Emory University; and Coulter Department research engineer Jorge Jimenez and undergraduate students Patrick Shannon and Dana Buice also contributed to this work.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Jul. 29, 2011
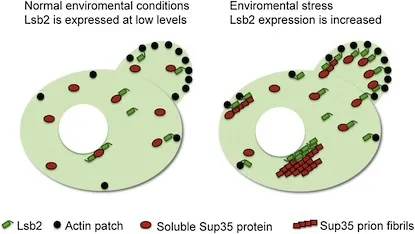
It’s achicken and egg question. Where do the infectious protein particles calledprions come from? Essentially clumps of misfolded proteins, prions causeneurodegenerative disorders, such as mad cow/Creutzfeldt-Jakob disease, inhumans and animals. Research in fungi has suggested that sometimes prions can alsohelp cells adapt to different conditions. Prions trigger the misfolding andaggregation of their properly folded protein counterparts, but they usuallyneed some kind of “seed” to get started.
Scientistshave studied a yeast protein called Lsb2 that can promote spontaneous prionformation. This unstable, short-lived protein is strongly induced by cellularstresses such as heat. Lsb2’s properties also illustrate how cells havedeveloped ways to control and regulate prion formation. The results arepublished in the July 22 issue of the journal Molecular Cell.
Thestudy was conducted by members of the Center for Nanobiology of the MacromolecularAssembly Disorders (NanoMAD) which is made up of scientists from the GeorgiaInstitute of Technology and Emory University. Scientists from the NationalInstitues of Health and the University of Illinois at Chicago also contributedto the study. The first author is senior associate Tatiana Chernova, PhD atEmory.
Theaggregated, or amyloid, forms of proteins connected with several otherneurodegenerative diseases such as Alzheimer’s, Parkinson’s and Huntington’scan, in some circumstances, act like prions. So the findings provide insightinto how the ways that cells deal with stress might lead to poisonous proteinaggregation in human diseases.
“Adirect human homolog of Lsb2 doesn’t exist, but there may be a protein thatperforms the same function,” said Keith Wilkinson, professor of biochemistry atEmory University School of Medicine. “The mechanism may say more about othertypes of protein aggregates than about classical prions in humans. Thismechanism of seeding and growth may be more important for aggregate formationin diseases such as Huntington’s.”
Lsb2does not appear to form stable prions by itself. Rather, it seems to bind toand encourage the aggregation of another protein, Sup35, which does formprions.
“Ourmodel is that stress induces high levels of Lsb2, which allows the accumulationof misfolded prion proteins,” Wilkinson said. “Lsb2 protects enough of thesenewborn prion particles from the quality control machinery for a few of them toget out.”
Incontinuation of previous research by Yury Chernoff, director of NanoMAD andprofessor in the School of Biology at Georgia Tech, the new data also show thatin addition to promoting new prions, Lsb2 strengthens existing prions duringstress.
"Littleis known about physiological and environmental conditions influencing amyloiddiseases in humans," said Chernoff. "Therefore, prophylacticmeasures, which could end up being more effective than therapies, areessentially non-existant. We hope that yeast model will help to fill thisgap."
Theresearch was supported by the National Institutes of Health.
Writtenby: Emory University and the Georgia Institute of Technology
News Contact
David Terraso
Georgia Tech College of Sciences
404-385-1393
Jul. 18, 2011
A new family of contrast agents that sneak into bacteria disguised as glucose food can detect bacterial infections in animals with high sensitivity and specificity. These agents -- called maltodextrin-based imaging probes -- can also distinguish a bacterial infection from other inflammatory conditions.
"These contrast agents fill the need for probes that can accurately image small numbers of bacteria in vivo and distinguish infections from other pathologies like cancer," said Niren Murthy, an associate professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University. "These probes could ultimately improve the diagnosis and treatment of bacterial infections, which remains a major challenge in medicine."
The imaging probes were described in the July 17, 2011 advance online edition of the journal Nature Materials. The research was sponsored by the National Science Foundation and National Institutes of Health.
Coulter Department postdoctoral fellows Xinghai Ning and Seungjun Lee led the project. University of Georgia Complex Carbohydrate Research Center postdoctoral associate Zhirui Wang; and Georgia State University Department of Biology associate professor Eric Gilbert and student Bryan Subblefield also contributed to the work.
In the United States in 2010, bacterial infections caused 40,000 deaths from sepsis and were the leading cause of limb amputations. A major limitation preventing the effective treatment of bacterial infections is an inability to detect them inside the body with accuracy and sensitivity. To image bacterial infections, probes must first deliver a large quantity of the contrast agent into bacteria.
"Most existing imaging probes target the bacterial cell wall and cannot access the inside of the bacteria, but maltodextrin-based imaging probes target a bacterial ingestion pathway, which allows the contrast agent to reach a high concentration within bacteria," said Murthy.
Maltodextrin-based imaging probes consist of a fluorescent dye linked to maltohexaose, which is a major source of glucose for bacteria. The probes deliver the contrast agent into bacteria through the organism's maltodextrin transporter, which only exists in bacterial cells and not mammalian cells.
"To our knowledge, this represents the first demonstration of a targeting strategy that can deliver millimolar concentrations of an imaging probe within bacteria," noted Murthy.
In experiments using a rat model, the researchers found that the contrast agent accumulated in bacteria-infected tissues, but was efficiently cleared from uninfected tissues. They saw a 42-fold increase in fluorescence intensity between bacterial infected and uninfected tissues. However, the contrast agent did not accumulate in the healthy bacterial microflora located in the intestines. Because systemically administered glucose molecules cannot access the interior of the intestines, the bacteria located there never came into contact with the probe.
They also found that the probes could detect as few as one million viable bacteria cells. Current contrast agents for imaging bacteria require at least 100 million bacteria, according to the researchers.
In another experiment, the researchers found that the maltodextrin-based probes could distinguish between bacterial infections and inflammation with high specificity. Tissues infected with E. coli bacteria exhibited a 17-fold increase in fluorescence intensity when compared with inflamed tissues that were not infected.
Additional laboratory experiments showed that the probes could deliver large quantities of imaging probes to gram-positive and gram-negative bacteria for internalization. Both types of bacteria internalized the maltodextrin-based probes at a rate three orders of magnitude faster than mammalian cells.
"Maltodextrin-based probes show promise for imaging infections in a wide range of tissues, with an ability to detect bacteria in vivo with a sensitivity two orders of magnitude higher than previously reported," said Murthy.
This project is supported by the National Science Foundation (NSF) (NSF Career Award No. BES-0546962) and the National Institutes of Health (NIH) (Award Nos. RO1 HL096796-01 and HHSN268201000043C). The content is solely the responsibility of the principal investigator and does not necessarily represent the official views of the NSF or NIH.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Vogel Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Vogel Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Jul. 18, 2011
Using a technique known as thermochemical nanolithography (TCNL), researchers have developed a new way to fabricate nanometer-scale ferroelectric structures directly on flexible plastic substrates that would be unable to withstand the processing temperatures normally required to create such nanostructures.
The technique, which uses a heated atomic force microscope (AFM) tip to produce patterns, could facilitate high-density, low-cost production of complex ferroelectric structures for energy harvesting arrays, sensors and actuators in nano-electromechanical systems (NEMS) and micro-electromechanical systems (MEMS). The research was reported July 15 in the journal Advanced Materials.
"We can directly create piezoelectric materials of the shape we want, where we want them, on flexible substrates for use in energy harvesting and other applications," said Nazanin Bassiri-Gharb, co-author of the paper and an assistant professor in the School of Mechanical Engineering at the Georgia Institute of Technology. "This is the first time that structures like these have been directly grown with a CMOS-compatible process at such a small resolution. Not only have we been able to grow these ferroelectric structures at low substrate temperatures, but we have also been able to pattern them at very small scales."
The research was sponsored by the National Science Foundation and the U.S. Department of Energy. In addition to the Georgia Tech researchers, the work also involved scientists from the University of Illinois Urbana-Champaign and the University of Nebraska Lincoln.
The researchers have produced wires approximately 30 nanometers wide and spheres with diameters of approximately 10 nanometers using the patterning technique. Spheres with potential application as ferroelectric memory were fabricated at densities exceeding 200 gigabytes per square inch -- currently the record for this perovskite-type ferroelectric material, said Suenne Kim, the paper's first author and a postdoctoral fellow in laboratory of Professor Elisa Riedo in Georgia Tech's School of Physics.
Ferroelectric materials are attractive because they exhibit charge-generating piezoelectric responses an order of magnitude larger than those of materials such as aluminum nitride or zinc oxide. The polarization of the materials can be easily and rapidly changed, giving them potential application as random access memory elements.
But the materials can be difficult to fabricate, requiring temperatures greater than 600 degrees Celsius for crystallization. Chemical etching techniques produce grain sizes as large as the nanoscale features researchers would like to produce, while physical etching processes damage the structures and reduce their attractive properties. Until now, these challenges required that ferroelectric structures be grown on a single-crystal substrate compatible with high temperatures, then transferred to a flexible substrate for use in energy-harvesting.
The thermochemical nanolithography process, which was developed at Georgia Tech in 2007, addresses those challenges by using extremely localized heating to form structures only where the resistively-heated AFM tip contacts a precursor material. A computer controls the AFM writing, allowing the researchers to create patterns of crystallized material where desired. To create energy-harvesting structures, for example, lines corresponding to ferroelectric nanowires can be drawn along the direction in which strain would be applied.
"The heat from the AFM tip crystallizes the amorphous precursor to make the structure," Bassiri-Gharb explained. "The patterns are formed only where the crystallization occurs."
To begin the fabrication, the sol-gel precursor material is first applied to a substrate with a standard spin-coating method, then briefly heated to approximately 250 degrees Celsius to drive off the organic solvents. The researchers have used polyimide, glass and silicon substrates, but in principle, any material able to withstand the 250-degree heating step could be used. Structures have been made from Pb(ZrTi)O3 -- known as PZT, and PbTiO3 -- known as PTO.
"We still heat the precursor at the temperatures required to crystallize the structure, but the heating is so localized that it does not affect the substrate," explained Riedo, a co-author of the paper and an associate professor in the Georgia Tech School of Physics.
The heated AFM tips were provided by William King, a professor in the Department of Mechanical Science and Engineering at the University of Illinois at Urbana-Champaign.
As a next step, the researchers plan to use arrays of AFM tips to produce larger patterned areas, and improve the heated AFM tips to operate for longer periods of time. The researchers also hope to understand the basic science behind ferroelectric materials, including properties at the nanoscale.
"We need to look at the growth thermodynamics of these ferroelectric materials," said Bassiri-Gharb. "We also need to see how the properties change when you move from the bulk to the micron scale and then to the nanometer scale. We need to understand what really happens to the extrinsic and intrinsic responses of the materials at these small scales."
Ultimately, arrays of AFM tips under computer control could produce complete devices, providing an alternative to current fabrication techniques.
"Thermochemical nanolithography is a very powerful nanofabrication technique that, through heating, is like a nanoscale pen that can create nanostructures useful in a variety of applications, including protein arrays, DNA arrays, and graphene-like nanowires," Riedo explained. "We are really addressing the problem caused by the existing limitations of photolithography at these size scales. We can envision creating a full device based on the same fabrication technique without the requirements of costly clean rooms and vacuum-based equipment. We are moving toward a process in which multiple steps are done using the same tool to pattern at the small scale."
In addition to those already mentioned, the research team included Yaser Bastani from the G.W. Woodruff School of Mechanical Engineering at Georgia Tech, Seth Marder and Kenneth Sandhage, both from Georgia Tech's School of Chemistry and Biochemistry and School of Materials Science and Engineering, and Alexei Gruverman and Haidong Lu from the Department of Physics and Astronomy at the University of Nebraska-Lincoln.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: John Toon (404-894-6986)(jtoon@gatech.edu) or Abby Robinson (404-385-3364)(abby@innovate.gatech.edu).
Writer: John Toon
News Contact
John Toon
Research News & Publications Office
Contact John Toon
404-894-6986
Jul. 11, 2011
A vaccine delivered to the skin using a microneedle patch gives better protection against the H1N1 influenza virus than a vaccine delivered through subcutaneous or intramuscular injection, researchers from Emory University and the Georgia Institute of Technology have found. Their research is published online in the Journal of Infectious Diseases.
Mice given a single H1N1 vaccine through the skin using a coated metal microneedle patch as well as mice vaccinated through subcutaneous injection were 100 percent protected against a lethal flu virus challenge six weeks after vaccination. However, when challenged with the H1N1 virus six months later, the injected mice had a 60 percent decrease in antibody production against the virus and extensive lung inflammation. Mice that were vaccinated with microneedles, on the other hand, maintained high levels of protection and antibody production after six months, with no signs of lung inflammation.
"A major goal of influenza vaccine development has been to confer strong immune responses, including immunological memory and cellular immune responses for long-term protection, and to limit virus spread after infection," said first author Dimitrios Koutsonanos, MD, post-doctoral fellow of microbiology and immunology at Emory University School of Medicine.
The research team also included Ioanna Skountzou, MD, PhD, Richard Compans, PhD, Maria del Pilar Martin, PhD, and Joshy Jacob, PhD, from Emory, and Georgia Tech bioengineers Mark Prausnitz, PhD, and Vladimir Zarnitsyn, PhD.
Researchers already have found that intramuscular injection is not the most efficient way to deliver vaccines. The muscles have a low concentration of cells needed to relay immune signals and activate a T-cell response, including dendritic cells, macrophages, and MHC class II-expressing cells. The skin, however, contains a rich network of antigen-presenting cells, including macrophages, Langerhans cells and dermal dendritic cells that activate cytokines and chemokines – immune signaling cells responsible for initiating an immune response.
The Emory/Georgia Tech research team previously reported that delivery of seasonal influenza vaccine through the skin using antigen-coated metal microneedle patches or dissolving microneedles elicited strong immune responses that can confer protection at least equal to conventional intramuscular injections. The team has developed dissolving microneedle technology that could be used in easy-to-administer, painless patches.
"The pandemic H1N1 A/California/04/09 influenza virus continues to be the predominant strain," said lead researcher Ioanna Skountzou, MD, PhD, assistant professor of microbiology and immunology at Emory University School of Medicine. "Our research shows that skin-based vaccination, made possible through microneedle technology, may now be a viable and more effective alternative to intramuscular injection for H1N1 flu and other strains as well."
"Microneedle delivery also offers other logistical advantages that make this method attractive for influenza vaccination, such as inexpensive manufacturing, small size for easy storage and distribution, and simple administration that might enable self-vaccination to increase patient coverage," said Prausnitz.
This news release was written by Emory University.
Media Contacts: Holly Korschun, Emory University (404-727-3990)(hkorsch@emory.edu) or John Toon (404-894-6986)(jtoon@gatech.edu).
News Contact
John Toon
Research News & Publications Office
Contact John Toon
404-894-6986
Jul. 06, 2011
Researchers at the Georgia Institute of Technology have designed a multiple-compartment gel capsule that could be used to simultaneously deliver drugs of different types. The researchers used a simple "one-pot" method to prepare the hydrogel capsules, which measure less than one micron.
The capsule's structure -- hollow except for polymer chains tethered to the interior of the shell -- provides spatially-segregated compartments that make it a good candidate for multi-drug encapsulation and release strategies. The microcapsule could be used to simultaneously deliver distinct drugs by filling the core of the capsule with hydrophilic drugs and trapping hydrophobic drugs within nanoparticles assembled from the polymer chains.
"We have demonstrated that we can make a fairly complex multi-component delivery vehicle using a relatively straightforward and scalable synthesis," said L. Andrew Lyon, a professor in the School of Chemistry and Biochemistry at Georgia Tech. "Additional research will need to be conducted to determine how they would best be loaded, delivered and triggered to release the drugs."
Details of the microcapsule synthesis procedure were published online on July 5, 2011 in the journal Macromolecular Rapid Communications.
Lyon and Xiaobo Hu, a former visiting scholar at Georgia Tech, created the microcapsules. As a graduate student at the Research Institute of Materials Science at the South China University of Technology, Hu is co-advised by Lyon and Zhen Tong of the South China University of Technology. Funding for this research was provided to Hu by the China Scholarship Council.
The researchers began the two-step, one-pot synthesis procedure by forming core particles from a temperature-sensitive polymer called poly(N-isopropylacrylamide). To create a dissolvable core, they formed polymer chains from the particles without a cross-linking agent. This resulted in an aggregated collection of polymer chains with temperature-dependent stability.
"The polymer comprising the core particles is known for undergoing chain transfer reactions that add cross-linking points without the presence of a cross-linking agent, so we initiated the polymerization using a redox method with ammonium persulfate and N,N,N’,N’-tetramethylethylenediamine. This ensured those side chain transfer reactions did not occur, which allowed us to create a truly dissolvable core," explained Lyon.
For the second step in the procedure, Lyon and Hu added a cross-linking agent to a polymer called poly(N-isopropylmethacrylamide) to create a shell around the aggregated polymer chains. The researchers conducted this step under conditions that would allow any core-associated polymer chains that interacted with the shell during synthesis to undergo chain transfer and become grafted to the interior of the shell.
Cooling the microcapsule exploited the temperature sensitivities of the polymers. The shell swelled with water and expanded to its stable size, while the free-floating polymer chains in the center of the capsule diffused out of the core, leaving behind an empty space. Any chains that stuck to the shell during its synthesis remained. Because the chains control the interaction between the particles they store and their surroundings, the tethered chains can act as hydrophobic drug carriers.
Compared to delivering a single drug, co-delivery of multiple drugs has several potential advantages, including synergistic effects, suppressed drug resistance and the ability to tune the relative dosage of various drugs. The future optimization of these microcapsules may allow simultaneous delivery of distinct classes of drugs for the treatment of diseases like cancer, which is often treated using combination chemotherapy.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Jun. 16, 2011
Stem cell technologies have been proposed for cell-based diagnostics and regenerative medicine therapies. However, being able to make stem cells efficiently develop into a desired cell type -- such as muscle, skin, blood vessels, bone or neurons -- limits the clinical potential of these technologies.
New research presented on June 16, 2011 at the annual meeting of the International Society for Stem Cell Research (ISSCR) shows that systematically controlling the local and global environments during stem cell development helps to effectively direct the process of differentiation. In the future, these findings could be used to develop manufacturing procedures for producing large quantities of stem cells for diagnostic and therapeutic applications. The research is sponsored by the National Science Foundation and the National Institutes of Health.
"Stem cells don't make any decisions in isolation; their decisions are spatially and temporally directed by biochemical and mechanical cues in their environment," said Todd McDevitt, director of the Stem Cell Engineering Center at Georgia Tech and an associate professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University. "We have designed systems that allow us to tightly control these properties during stem cell differentiation, but also give us the flexibility to introduce a new growth factor or shake the cells a little faster to see how changes like these affect the outcome."
These systems can also be used to compare the suitability of specific stem cell types for a particular use.
"We have developed several platforms that will allow us to conduct head-to-head studies with different kinds of stem cells to determine if one type of stem cell outperforms another type for a certain application," said McDevitt, who is also a Petit Faculty Fellow in the Institute for Bioengineering and Bioscience at Georgia Tech.
Many laboratory growth methods allow stem cells to aggregate in three-dimensional clumps called "embryoid bodies" during differentiation. McDevitt and biomedical engineering graduate student Andres Bratt-Leal incorporated biomaterial particles directly within these aggregates during their formation. They introduced microparticles made of gelatin, poly(lactic-co-glycolic acid) (PLGA) or agarose and tested their impact on the assembly, intercellular communication and morphogenesis of the stem cell aggregates under different conditions by varying the microsphere-to-cell ratio and the size of the microspheres.
The researchers found that the presence of the biomaterials alone modulated embryoid body differentiation, but did not adversely affect cell viability. Compared to typical delivery methods, providing differentiation factors -- retinoic acid, bone morphogenetic protein 4 (BMP4) and vascular endothelial growth factor (VEGF) -- via microparticles induced changes in the gene and protein expression patterns of the aggregates.
By including tiny magnetic particles into the embryoid bodies during formation, the researchers also found they could use a magnet to spatially control the location of an aggregate and its assembly with other aggregates. The magnetic particles remained entrapped within the aggregates for the duration of the experiments but did not adversely affect cell viability or differentiation.
"With biomaterial and magnetic microparticles, we are beginning to be able to recreate the types of complex geometric patterns seen during early development, which require multiple cues at the same time and the ability to spatially and temporally control their local presentation," noted McDevitt.
While microparticles can be used to control differentiation by regulating the local environment, other methods exist to control differentiation through the global environment. Experiments by McDevitt and biomedical engineering graduate student Melissa Kinney have demonstrated that modulating hydrodynamic conditions can dictate the morphology of cell aggregate formation and control the expression of differentiated phenotypic cell markers.
"Because bioreactors typically impose hydrodynamic forces on cells to cultivate large volumes of cells at high density, our use of hydrodynamics to control cell fate decisions represents a novel, yet simple, principle that could be used in the future for the scalable efficient production of stem cells," added McDevitt.
Technologies capable of being directly integrated into bioprocessing systems will be the best choice for manufacturing large batches of stem cells, he noted. In the future, the development of multi-scale techniques that combine different levels of control -- both local and global -- to regulate stem cell differentiation may help the translation of stem cells into viable clinical therapies.
This project is supported by the National Science Foundation (NSF) (Award No. CBET 0651739) and the National Institutes of Health (NIH) (R01GM088291). The content is solely the responsibility of the principal investigator and does not necessarily represent the official views of the NSF or NIH.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
Contact Abby Robinson
404-385-3364
Pagination
- Previous page
- Page 45
- Next page