Apr. 08, 2025
Georgia Tech School of Electrical and Computer Engineering (ECE) Professor Christopher Rozell was inducted into the American Institute for Medical and Biological Engineering (AIMBE) College of Fellows at the AIMBE Annual Event on March 31 in Arlington, Va.
College membership honors those, “who have made outstanding contributions to engineering and medicine research, practice, or education,” and “to the pioneering of new and developing fields of technology, making major advancements in traditional fields of medical and biological engineering or developing/implementing innovative approaches to bioengineering education.”
The distinction is among the highest professional distinctions given to medical and biological engineers, comprised of the top two percent of engineers in these fields.
He was nominated and inducted for outstanding contributions to computational neuroengineering, psychiatric neuromodulation, and international leadership in accessible biomedical education.
Rozell's research interests are in computational neuroengineering, an intersection of neuroscience, data science, neurotechnology and computational modeling that advances the understanding of brain function and the design of effective interventions.
His research has a particular focus on advancing our understanding and novel brain stimulation therapies for psychiatric disorders such as treatment resistant depression.
Recently, he was part of a team that identified a unique pattern in brain activity that reflects the recovery process in patients with treatment-resistant depression. This pattern, known as a biomarker, represented a significant advance in treatment for the most severe and untreatable forms of depression.
His work also includes research that takes a creative approach to advancing the understanding of the societal impacts of emerging technologies such as neurotechnology and AI.
Rozell especially takes pride in being a first-generation scholar who is committed to accessibility in scientific communities. In pursuit of this goal, he co-founded and serves on the Board of Directors of Neuromatch, Inc., a global nonprofit increasing access to scientific knowledge.
His scholarly efforts have resulted in many published works in top publications, such as Nature, and a number of awards, including the NSF CAREER Award.
Before joining the ECE faculty in 2008 as an assistant professor, Rozell received a B.S.E. degree in computer engineering and a B.F.A. degree in music in 2000 from the University of Michigan. He then received M.S. and Ph.D. degrees in electrical engineering in 2002 and 2007 from Rice University and was a postdoctoral scholar at the Redwood Center for Theoretical Neuroscience at the University of California, Berkeley.
News Contact
Zachary Winiecki
Mar. 26, 2025
Georgia Tech stands on the brink of a medical revolution, fueled by a monumental award from the Marcus Foundation. This transformative $40 million endeavor, with a principal investment of $20 million from the Marcus Foundation, promises to make high-quality, life-saving cell therapies more affordable, reliable, and accessible than ever before.
This was among the final initiatives personally directed by Bernie Marcus, the philanthropist, entrepreneur, and The Home Depot co-founder, before his passing in November 2024. Marcus invited Georgia Tech President Ángel Cabrera to his home in Boca Raton, Florida, to discuss Georgia Tech’s capability to usher in a new era of regenerative medicine.
“I’ll never forget my conversation with Bernie,” Cabrera said. “His challenge to Georgia Tech was clear: Use our engineering expertise to make cell therapies more accessible and cost-effective and develop cures for incurable diseases.
“This generous award is a testament to our shared belief in the power of innovation and technology to improve lives, and it’s an honor for Georgia Tech to fulfill Bernie’s vision for the future of healthcare,” he added.
The funding will ignite innovation at Georgia Tech’s Marcus Center of Excellence for Cell Biomanufacturing, formerly named the Marcus Center for Therapeutic Cell Characterization and Manufacturing, which has been bioengineering potential cellular cures for more than seven years. It will enable Georgia Tech engineers to advance work at the center and within the National Science Foundation-funded Engineering Research Center in Cell Manufacturing Technologies (CMaT), to develop automated bioreactor systems that eliminate the need for costly cleanrooms.
Marcus/CMaT Director Johnna Temenoff compared the current state of cell therapies to the early days of the automobile industry. She explained this new injection of funds will allow her team to shift from handcrafted production to an assembly-line approach.
“I firmly believe that for us to make good on the promises of these biotechnologies to improve healthcare worldwide, we must be able to manufacture them in a more reproducible and cost-effective manner. Georgia Tech’s distinctive strength lies in our engineering expertise, allowing us to tackle difficult biological problems,” Temenoff said.
The impact of this award extends beyond the laboratory. It has the potential to significantly boost Georgia's bioeconomy, making the state a hub for advanced therapy development and biomanufacturing. It will attract jobs and top-tier talent to the region.
Dr. Jonathan Simons, chief science officer and medical director of the Marcus Foundation, said Bernie Marcus liked to think of cells as “living drugs.”
Simons explained, “This is life-extending, lifesaving, and life-changing material. It's not like making a drug like penicillin or Tylenol. This is not like a little blister pack of pills. This is a whole new frontier for pharmacology and the pharmaceutical industry.”
Simons emphasized this is the latest chapter of both the Marcus Foundation’s investment in biomedical engineering at Georgia Tech and Bernie Marcus’s enduring biomedical research philanthropy.
“I think Bernie would say, ‘I’m not interested in my legacy. I’m interested in how many patients in five years will benefit from this $40 million effort. It’s all about lives changed, lives saved, and diseases ended,’” he said.
To learn more about Georgia Tech’s research in cell and gene therapy biomanufacturing, visit cellmanufacturing.gatech.edu.
News Contact
Shelley Wunder-Smith
Director of Research Communications
Mar. 19, 2025
Biomedical engineers at Georgia Tech have developed a system for collecting and processing thermal images that allows for reliable, detailed measurement of vital signs such as respiration and heart rate or body temperature.
Their monitoring approach is passive and requires no contact. The system could one day lead to early detection for cancer or other diseases by flagging subtle changes in body tissues.
The researchers have overcome the spectral ambiguity inherent in conventional thermal imaging, sharpening the texture and detail they can extract from images and removing the effects of heat from the environment surrounding a subject. They published details of their work March 19 in the journal Cell Reports Physical Science.
News Contact
Joshua Stewart
College of Engineering
Mar. 19, 2025
Georgia Tech scientists are revealing how decades-long research programs have transformed our understanding of evolution, from laboratory petri dishes to tropical islands — along the way uncovering secrets that would remain hidden in shorter studies.
Through a new review paper published in Nature, the researchers underscore how long-term studies have captured evolution's most elusive processes, including the real-time formation of new species and the emergence of biological innovations.
"Evolution isn't just about change over millions of years in fossils — it's happening all around us, right now," says James Stroud, the paper’s lead author and an Elizabeth Smithgall Watts Early Career Assistant Professor in the School of Biological Sciences at Georgia Tech. "However, to understand evolution, we need to watch it unfold in real time, often over many generations. Long-term studies allow us to do that by giving us a front-row seat to evolution in action."
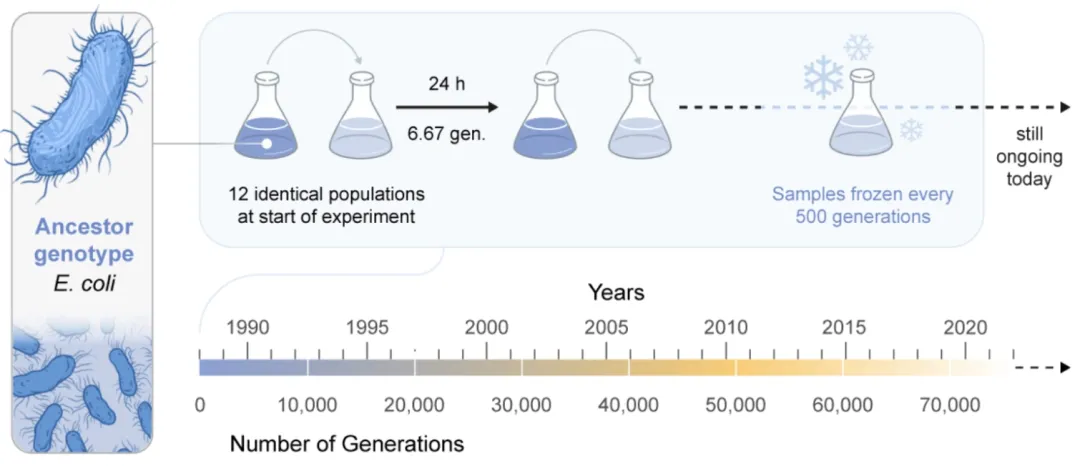
The paper, “Long-term studies provide unique insights into evolution,” is the first-ever comprehensive analysis of these types of long-term evolutionary studies, and examines some of the longest-running evolutionary experiments and field studies to date, highlighting how they provide new perspectives on evolution. For example, in the Galápagos, a 40-year field study of Darwin’s finches — songbirds named after evolutionary biology’s famous founder — documented the formation of a new species through hybridization. In the lab, a study spanning 75,000 generations of bacteria showed populations unexpectedly evolving completely new metabolic abilities.
“These remarkable evolutionary events were only caught because of the long-term nature of the research programs,” Stroud says. “Even if short-term studies captured similar events, their evolutionary significance would be hard to assess without the historical context that long-term research provides.”
“The most fascinating results from long-term evolution studies are often completely unexpected — they're serendipitous discoveries that couldn't have been predicted at the start,” explains the paper’s co-author, Will Ratcliff, Sutherland Professor in the School of Biological Sciences and co-director of the Interdisciplinary Ph.D. in Quantitative Biosciences at Georgia Tech.
“While we can accelerate many aspects of scientific research today, evolution still moves at its own pace,” Ratcliff adds. “There's no technological shortcut for watching species adapt across generations.”
Decades of discovery — from labs to islands
The new paper also highlights a growing challenge in modern science: the critical importance of supporting long-term research in an academic landscape that increasingly favors quick results and short-term funding. Yet, they say, some of biology's most profound insights emerge only through multi-decadal efforts.
Those challenges and rewards are familiar to Stroud and Ratcliff, who operate their own long-term evolutionary research programs at Georgia Tech.
In South Florida, Stroud’s ‘Lizard Island’ is helping document evolution in action across the football field-sized island’s 1,000-lizard population. By studying a community of five species, his research is providing unique insights into how evolution maintains species’ differences, and how species evolve when new competitors arrive. Now operating for a decade, it is one of the world’s longest-running active evolutionary studies of its kind.
In his lab at Georgia Tech, Ratcliff studies the origin of complex life — specifically, how single-celled organisms become multicellular. His Multicellularity Long Term Evolution Experiment (MuLTEE) on snowflake yeast has run for more than 9,000 generations, with aims to continue for the next 25 years. The work has shown how key steps in the evolutionary transition from single-celled organisms to multi-celled organisms occur far more easily than previously understood.
Important work in a changing world
Stroud says that the insights from these types of studies, and this review paper, are arriving at a crucial moment. “The world is rapidly changing, which poses unprecedented challenges to Earth's biodiversity,” he explains. “It has never been more important to understand how organisms adapt to changing environments over time.”
“Long-term studies provide our best window into achieving this,” he adds. “We can document, in real time, both short-term and long-term evolutionary responses of species to changes in their environment like climate change and habitat modification."
By drawing together evolution's longest-running experiments and field studies for the first time, Stroud and Ratcliff offer key insights into studying this fundamental process, suggesting that understanding life's past — and predicting its future — requires not just advanced technology or new methods, but also the simple power of time.
Funding: The US National Institutes of Health and the NSF Division of Environmental Biology
Mar. 18, 2025
In the movies, Ant-Man can shrink down to the size of an insect to carry out his superhero missions. It makes for fun cinema, but of course, it is impossible. For starters, biological systems can’t scale up or down and stay proportional. The hero would die before throwing his first teeny, tiny punch.
That’s miniaturization science for you. It’s the study of how materials and systems behave at microscopic scales, and it’s transforming biomedical engineering. And though it has led to breakthroughs in diagnostics and treatments, “teaching students about the subject is really challenging,” said David Myers, assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory.
“It’s because the behavior of fluids and materials at such small scales defies intuition, and you can’t really observe what’s going on,” added Myers, who understands the instructional challenge well — he teaches a graduate level course focused on translational microsystems, which is heavily integrated with his lab’s research.
Recognizing the limitations of traditional coursework, Myers and his collaborators have developed a different approach. In Myers’ class, students build and test and observe the workings of microfluidic devices, a hallmark of miniaturization science — microfluidics is the manipulation of tiny volumes of fluids in miniaturized devices.
Their new approach has made all the difference, even earning Myers a CIOS Award for teaching excellence. But Myers is quick to emphasize that this was a team effort. He and his lab developed a hands-on activity to help students learn device construction (and the underlying technical concepts).
Then he reached out to Todd Fernandez, senior lecturer and Coulter BME’s director of learning innovation. Together they optimized the activity to maximize students’ learning. That has evolved into an ongoing partnership between technical and educational research faculty in the department, resulting in an article in the journal Lab on a Chip.
"In other microfluidics courses, you walk through the step-by-step process of fabrication, but actually seeing the device come together in front of you provides such valuable insight into the underlying concepts and manufacturing techniques,” explained Priscilla Delgado, a fifth-year graduate student in Myers’ lab and lead author of the published study. “That hands-on experience is crucial for truly understanding this technology."
Bridging Critical Gaps
Myers’ course bridges several critical gaps, including the high cost of advanced learning activities. It also addresses student misconceptions.
“The primary objective isn’t just the successful construction of devices, but a deeper conceptual understanding of miniaturization science and design principles,” said Myers, whose approach emphasizes conceptual change.
Students often come into the course with misunderstandings about microscale phenomena, “assuming that fluid flow at this scale behaves the same way as in larger systems,” Myers said.
Delgado added, “but it’s wild how fluid behavior changes at the microscale. If you mix two colored liquids in a regular cup, you get a third color. But in microfluidics, the laminar flow and reliance on diffusion can keep those streams separate — it really challenges your intuition about mixing.”
The class allows students to build and test microfluidic kits — mixers, valves, and bubble generators, using inexpensive, widely available materials. This activity is structured to help students encounter misunderstandings and work through them. Rather than simply presenting correct information, instructors guide students through a learning cycle in which they identify errors, reflect on their mistakes, and refine their understanding.
“You can see their brains just sizzle,” said Myers. “Then you kind of add a little bit of structure. You ask, ‘Are you sure you have all the layers there that you’re thinking about?’ And then they’ll go back, count, and realize—oh, there’s this missing middle layer.”
The layer-by-layer assembly technique uses laser-cut adhesive films to construct microfluidic devices. Because the devices are assembled from transparent layers, students can see how their designs function and they can troubleshoot any errors.
“One of the best things about these sticker-based microfluidic devices is how easy they are to prototype,” said Delgado. “I can literally have a new design laser-cut and assembled within an hour, rather than waiting months using traditional methods. The accessibility and speed of iteration is a game-changer."
Expanding the Possibilities
Beyond its accessibility, the sticker-based microfluidic approach also expands the possibilities for innovation.
“The really cool thing is, this is a sticker,” Myers said. “You can place it on your skin. You can place it on the table. You can place it on the wall, if you really felt like it. And when you integrate it with high-end instrumentation like advanced sensors, suddenly you have a resource that traditional microfluidics can’t easily replicate.”
This kind of flexibility enables students to explore microfluidics in new ways. The study involved 57 students, some of whom took their designs beyond the classroom.
“I cannot say enough how much I love how accessible it is and the portability of it,” Delgado said. “You can do this anywhere. You could do this at home. We’ve done it at science fairs for high school students to really challenge the way they think about mixing.”
The impact of the work has also influenced the direction Delgado wants to take in her career. She’s found herself drawn deeper into the field, inspired by microfluidic design.
“The first time I laid eyes on that microfluidic device I had just built, I was captivated,” she said. “I remember thinking, ‘This is so cool; I have to dive deeper into this field.’ That’s when I knew a PhD was in my future, even though I had initially planned otherwise.”
This approach to teaching miniaturization science not only enhances learning but also democratizes access to innovation, according to Myers.
“The really cool thing that I love about this activity is that you’re sharing knowledge and power with the people using the technology,” he said. “Instead of them receiving technology from some high-resource institution, they’re able to look at the problems and start addressing them themselves.”
Miniaturization science plays a crucial role in developing point-of-care medical devices and other low-cost diagnostic tools, particularly in resource-limited settings. Equipping students around the world with the ability to create microfluidic systems could help empower future researchers and engineers.
Fernandez believes this hands-on approach represents a shift in how miniaturization science will be taught.
“By focusing on student-driven exploration and conceptual understanding rather than rote device assembly, educators can better prepare the next generation of engineers and scientists to navigate and contribute to the ever-expanding world of microsystems,” he said. “ And what’s really cool is, you let them play, and they learn more. They discover things that we didn’t even have time to teach them.”
News Contact
Mar. 18, 2025
In 2023 the Wallace H. Coulter Department of Biomedical Engineering launched a new program designed to train the next generation of leaders in cardiovascular research. Five first-year graduate students formed the first cohort that fall.
Currently, there are nine students in the Cardiovascular Biomechanics Graduate Training Program at Emory and Georgia Tech (CBT@EmTech). The program offers two years of training in an assortment of disciplines, including cardiovascular biomechanics, mechanobiology, medical imaging, computational modeling, medical devices, therapeutics discovery and delivery, and data science.
“The goal of the program is to stimulate interdisciplinary training,” so we expose the students to multiple areas of research,” says Hanjoong Jo, CBT@EmTech director, Wallace H. Coulter Distinguished Professor.
“And we have a very diverse group of trainees interested in various aspects of cardiovascular research and medicine,” Jo added. “Four out of five students from our first cohort already have secured prestigious fellowships, demonstrating the caliber of the trainees in the program.”
The students from that cohort brought a wide range of experiences, interests, and ambitions to the program. Now in their final months as CBT@EmTech trainees, they took time to share their stories.
Yohannes Akiel
Principal Investigator: Michael Davis
Campus: Emory
Undergraduate: University of Texas-San Antonio
I've always had a passion for helping people and I feel that I’m doing this through my research on aortic valve tissue engineering for pediatric patients. Aortic valve disease is found in 1-2% of live births, because of congenital heart defects or infections. Current valve replacements are limited — for one thing, they’re incapable of growing and remodeling with the patient. This presents a need for a new tissue-engineered valve that can address these challenges. In the Davis lab, we’re working on a tissue engineered heart valve to provide a better, long-term solution.
Throughout my time in the CBT@EmTech program, I've gained a range of knowledge in the cardiovascular space, learning about atherosclerosis, peripheral artery disease, valve disease, as well as computational and imaging techniques to help solve some of these problems. As part of the program, we are also required to take an Advanced Seminar class in the cardiovascular area.
Through this class, I was able to participate in some interesting clinical observations in the Emory University Hospital cardiology department. For example, I watched a cardiologist perform a transesophageal echocardiogram. The doctor was checking for heart blockages on a patient who had atrial fibrillation. This procedure was followed by a cardioversion to restore a normal heart rhythm. This was a profound demonstration of biomedical technology in action that left a lasting impression on me.
Leandro Choi
Principal Investigator: Hanjoong Jo
Campus: Emory
Undergraduate: Duke University
As a PhD student in the Jo Lab, I am studying how disturbed flow influences transcriptional regulation in endothelial cell reprogramming and atherosclerosis. Our goal is to identify and develop therapeutics that target non-lipid residual pathways contributing to this widespread and deadly disease.
I initially became interested in this line of research due to a family history of cardiovascular disease. As an undergraduate, I worked in a tissue engineering lab where I employed stem cell and tissue engineering methods to model the circulatory system. A desire to further explore the role of mechanosensitive genes and proteins in cardiovascular disease led me to pursue a PhD in this field.
One of the most valuable aspects of the CBT@EmTech program has been the opportunity to connect with a network of students and faculty who are leaders in cardiovascular research. Through monthly meetings, we share our work and gain insights into the diverse engineering applications our interdisciplinary program brings to the field, with the common goal of improving cardiovascular health.
Aniket Venkatesh
Principal Investigator: Lakshmi Prasad
Campus: Georgia Tech
Undergraduate: Georgia Tech
October 2024 marked the three-year anniversary of my uncle’s passing due to complications from a mild heart attack. His angiogram showed 30% vessel blockage, leading to heart surgery. Sadly, he suffered a brain stroke days later, resulting in deteriorating speech, muscle movement, and eventually death at 48. This personal tragedy brought urgency to my research questions: Can the risk of complications following cardiovascular treatments be predicted? Can underlying cardiovascular pathology be treated before it progresses to a heart attack or stroke? Was my uncle’s death preventable? These questions drive my cardiovascular research, focused on predicting post-procedural heart valve outcomes through computational modeling.
Being part of the prestigious CBT@EmTech program at Emory and Georgia Tech has significantly advanced my research journey. Learning from fellow trainees, presenting my research, and attending academia-focused workshops (like one about grant writing) have helped me stand out in heart valve computational modeling. The program, along with my PI, Dr. Lakshmi Prasad Dasi, and co-PI, Dr. John Oshinski, has provided the resources needed to translate my research from the lab to the clinic through regular meetings with clinicians and data transfer to and from hospitals. I am grateful for the opportunity to pursue my long-term goal of predicting risks of complications before cardiovascular treatments and helping prevent adverse clinical outcomes like those experienced by my uncle.
Isabel Wallgren
Principal Investigator: Simone Douglas-Green
Campus: Georgia Tech
Undergraduate Degree: University of Virginia
Peripheral artery disease (PAD) occurs when atherosclerotic plaque accumulates in limb arteries, blocking blood flow. Current interventions limit disease progression, but surgery is often needed to prevent critical limb ischemia. A less invasive approach promotes angiogenesis and arteriogenesis to strengthen collateral vessels and bypass blockages. The Hansen Lab studies satellite cells (SCs), which repair muscle fibers and release growth factors, as a potential PAD therapy.
My research focuses on improving the delivery of SCs using a special fibrin scaffold in a mouse model of blocked blood flow in the legs. By adjusting the properties of the fibrin scaffold, we can create an environment that helps these cells grow and renew themselves. We study how quickly the fibrin forms to ensure the cells stay where we inject them and how it breaks down to keep a steady supply of renewing SCs. We believe that with fibrin, the cells will move into the damaged tissue, repair muscle fibers, and release growth factors to encourage new blood vessel growth.
The goal is to create alternative treatments for PAD that prevent disease progression and improve patients' quality of life.
The CBT@EmTech program has given me a supportive network of peers and mentors, enhancing my growth as a researcher. The program chairs have tailored the curriculum to our needs and allowed us to shape it. For example, I’ve had the privilege of co-planning our biannual retreat. We recruited guests for two panels and invited a guest speaker for a storytelling workshop. This retreat shows how the program imparts knowledge beyond research, aiming to improve our scientific storytelling and self-presentation skills, valuable for any career.
Deborah Wood
Principal Investigator: Simone Douglas-Green
Campus: Georgia Tech
Undergraduate Degree: University of Virginia
As a researcher, I am challenged to explore the unknown. Moreover, my role as an engineer is rooted in using knowledge that has already been conceptualized. Combining these perspectives as a biomedical engineer has led me to pursue research with an emphasis on improving human health.
Today, cardiovascular diseases represent the global leading cause of death. While this glaring statistic indicates the egregious burden of cardiovascular diseases, my parents' lived experiences with cardiovascular diseases is what drives me to use my life’s work to address critical challenges at the intersection of the cardiovascular field and biomedical engineering.
My research seeks to alleviate cardiovascular diseases by using nanoparticles to target endothelial cells, which line the innermost layer of blood vessels and contribute to blood vessel function. The Cardiovascular Biomechanics and Mechanobiology Program at Emory (CBT@EmTech) has given me an avenue to pursue this research.
Through my CBT@EmTech co-mentorship, I have developed a foundation in endothelial cell biology and atherosclerosis. I have also been challenged to think critically about how my research benefits both science and society through my exposure to prominent cardiovascular researchers. My experiences with CBT@EmTech have made me eager to use my training to pursue a postdoc in the and eventually lead a lab answering critical questions in cardiovascular research.
News Contact
Mar. 14, 2025
Is there a tried-and-true formula to drive achievement in the corporate world? For many College of Sciences alumni, the surprising answer lies in science fundamentals — particularly the scientific method.
We spoke to three alumni about the benefits of applying a scientific approach to business.
Navigating the Startup Landscape
Thomas Kim graduated from Georgia Tech in 1992 with a bachelor’s degree in chemistry, intending to pursue a career in academia. Instead, after earning a master’s in biochemistry and a law degree, then working as a biotech attorney, he is now president and CEO of two life science startups.
“The entire startup company process can be construed as an exercise in the scientific method,” says Kim. “In the early stage, you start with preclinical data and a thesis on how that translates to human disease. Next, you pressure test everything. Depending on confidence in your results, you continue to invest and move the program forward to translate your initial idea into a potential human therapeutic, or you pivot to a different application or drug in the pipeline.”
One of his current companies, Epivario aims to develop treatments for preventing relapse in drug and alcohol addiction and PTSD.
“We’re in the preclinical development stage, requiring constant testing – and retesting. It’s an arduous, ongoing task where not everything works the first time – or the 50th.”
In the fast-moving start-up world, decisions must be made quickly and, most importantly, accurately to stay ahead of the competition. Kim points to a background in the scientific method as foundational to making crucial business decisions. “Whether you’re responsible for research and development or company strategy, it’s a key skill to take deep analysis and translate it into quality decision making.”
On a broader level, Kim admits he sees his work more as a mission than a job.
“I feel fortunate to work in a field where our efforts can improve human lives.”
From Lab to Leadership
After graduating with a bachelor’s degree in microbiology from Washington State University, Maureen Metcalfe (M.S. BIO 2014) scored her dream job as a CDC electron microscopist in 2007, then enrolled part-time at Georgia Tech to earn a master’s in biology. As part of her master’s requirements, she also conducted research in Professor Ingeborg Schmidt-Krey’s laboratory, where she attempted to create conditions to crystallize a protein involved in Alzheimer's pathogenesis. Between her full-time job, academic studies, and work in the laboratory, she averaged more than 70 hours of work each week.
“I lived the scientific method – especially the test your hypothesis part,” says Metcalfe. “Over four years, I had 600 failures.”
Those failures taught her resilience and time management – skills vital to her current consulting career.
“It’s more ingrained than step by step, but almost every time there is a problem on a client project, I rely on certain aspects of the scientific method,” says Metcalfe. "I first observe, research, and analyze the data, re-tool if necessary, and then apply that data to make an informed recommendation to the client.”
Over the years, the perseverance she developed in the laboratory has helped her push on to complete complicated client projects.
“I think the scientific process and what it gives us is unique,” says Metcalfe. “Science gives you the skill set to keep asking questions and not accept a failure or setback.”
Metcalfe can even apply aspects of her career trajectory to principles inherent in the scientific method.
“Building on what you learn and changing course is inherent in the scientific method. I realized I wanted different challenges in my life, and I left a career in government to find them. Taking my science degree into new work situations has been very gratifying. The foundation I built in science serves me well in the challenging, fast-paced, and exciting world of consulting.”
Building Career Success
A night out with friends upended and redirected Christa Sobon’s carefully constructed career plans. After earning psychology and history degrees with a minor in French from Emory University, Sobon, (M.S. PSY 1996) came to Georgia Tech to build a career in academia. Those plans changed when she talked to a friend’s wife at a party who told her that Accenture liked to hire smart people who could solve problems.
After two years at Tech in a quantitative program focused on methodology and research seeped in the scientific method, Sobon was confident of her problem-solving abilities. Forgoing academia, she accepted a job at Accenture and has spent more than 29 years leading programs that drive business success at companies including All Connect, Netspend, and Jabian Consulting. Currently, she is operations management senior director at Cox Automotive.
“I’ve been able to use elements of the scientific method in every place I’ve worked,” says Sobon. “The scientific method equips you with critical thinking skills and promotes a methodical approach to tackling challenges that works well in the corporate world.”
As a program manager for most of her career, she cites forming a hypothesis and analyzing the data as the most critical steps when figuring out how to get a product to market.
“We gather data in terms of understanding the customer pain points, then form the hypothesis (or in our case a new product) designed to solve that particular problem. When we believe we have a workable solution, we bring that product to market,” says Sobon.
She explains that they rarely stick the landing on the first try.
“I’ve led teams where we were convinced the customer would love our product…when the customer did NOT love our product, we would then refine, test in the market again, and continue to iterate until we launched a successful product – basically a mini-version of the scientific method.”
Sobon is a strong believer in a scientific education – and Georgia Tech.
“The rigor that you learn at Georgia Tech about approaching problem-solving through the scientific method has so many applications. These skills are transferable across a variety of fields and enable individuals to analyze complex problems, develop innovative solutions, and make data-driven decisions, all of which are essential in business today.”
News Contact
Laura S. Smith, Writer
Mar. 12, 2025
Shriners Children’s is a pediatric healthcare system for orthopedic, spine, and burn injuries, as well as other specialty care and rehabilitation. Shriners provides treatment for nearly 20,000 children and families from over 130 countries around the world with one mission: compassionate, innovative care that improves the quality of life for children and their families. As part of its research mission, Shriners Children's collaborates with academic and industry partners to develop leading tools, processes, and programs that improve pediatric care.
Georgia Tech’s Pediatric Innovation Network (PIN) has collaborated with Shriners Children’s since 2017. The initiative connects researchers — including engineers, data analysts, scientists, and others — with frontline pediatric clinicians to create new technologies for unmet pediatric healthcare needs. This dynamic collaboration began with a conversation between Marc Lalande, vice president of research at Shriners’ Children’s, and Leanne West, chief engineer of pediatric technologies at Georgia Tech, who were introduced by a mutual colleague.
That conversation has resulted in 22 projects to date, spanning topics such as artificial intelligence, data management, robotic exoskeletons, augmented reality games, wearable sensors, and more. The collaboration between Shriners Children’s and GT PIN works to engage faculty and students within Georgia Tech’s cutting-edge research ecosystem and multiple hospitals within Shriners’ network.
“From the very beginning, this has been an amazing collaboration. Our faculty love working on the real-world problems brought to us by Shriners’ clinicians,” said West.
Learn more about one of these projects on how virtual reality is transforming assessment of patients with upper limb movement challenges.
News Contact
Savannah Williamson
Communications Manager, IBB
Mar. 05, 2025
A multidisciplinary team of researchers at Georgia Tech has discovered how lateral inhibition helps our brains process visual information, and it could expand our knowledge of sensory perception, leading to applications in neuro-medicine and artificial intelligence.
Lateral inhibition is when certain neurons suppress the activity of their neighboring neurons. Imagine an artist drawing, darkening the lines around the contours, highlighting the boundaries between objects and space, or objects and other objects. Comparably, in the visual system, lateral inhibition sharpens the contrast between different visual stimuli.
“This research is really getting at how our visual system not only highlights important things, but also actively suppresses irrelevant information in the background,” said lead researcher Bilal Haider, associate professor in the Wallace H. Coulter Department of Biomedical Engineering. “That ability to filter out distractions is crucial.”
Understanding how these inhibitory mechanisms work could provide insights into why people have trouble filtering out distractions or focusing on what’s important, in conditions like autism or ADHD.
“Our findings may also influence how we design artificial intelligence and neural networks,” said Haider, whose team published its work this month in Nature Neuroscience. “Current AI systems treat all the computing units the same, but the brain has figured out how to assign specialized computing roles.”
Joseph Del Rosario, a former graduate student in the Haider lab, was the lead author. Another key contributor was Hannah Choi, assistant professor in the School of Mathematics, and her Research Group in Mathematical Neuroscience. Their team built computational models to test the biological findings.
“Collaborating with mathematicians to really understand the computational principles underlying these inhibitory processes is a great example of how neuroscience can inform fields like AI,” Haider said.
Read more in the Coulter Department of Biomedical Engineering newsroom.
Feb. 24, 2025
Four million Americans suffer from glaucoma, an incurable eye disease that slowly degrades peripheral vision and eventually leads to blindness. Researchers at Georgia Tech have discovered a potential way to stop this degradation and possibly save people’s vision before it’s too late.
Raquel Lieberman, a professor in the School of Chemistry and Biochemistry and the Parker H. Petit Institute for Bioengineering and Bioscience, and her lab team have discovered two new antibodies with promise to treat glaucoma. The antibodies can break down the protein myocilin, which, when it malfunctions, can cause glaucoma.
Lieberman’s group recently published this research in the Proceedings of the National Academy of Sciences: Nexus.
Protein Problems
Myocilin is just one of hundreds of thousands of proteins that make up the human body. In the eye, an especially delicate balance of proteins and fluid enables sight. The aqueous humor, a clear fluid, bathes the lens that helps focus light into the retina. In a healthy eye, the fluid drains regularly, but if something prevents the fluid from circulating, it increases pressure.
“Your eyeball is kind of like a basketball,” explained Lieberman. “If you want it to work optimally, it has to be pressurized.”
Lieberman’s team has learned that if myocilin mutates, it clumps up and prevents aqueous humor from draining, increasing eye pressure. If left unmanaged, glaucoma and — eventually — blindness will occur.
Antibody Answer
Lieberman’s lab characterized two new antibodies that each, in their unique way, can destroy myocilin gone rogue. One binds in a way that does not prevent myocilin from clumping; the other prevents the protein from aggregating. Both effectively break down myocilin so it no longer blocks the aqueous humor from flowing.
“These exciting results provide proof of concept that targeted antibodies for mutant myocilin aggregation could be therapeutic,” said Alice Ma, a Ph.D. graduate who worked on the research. “This represents a new paradigm for treating other diseases associated with protein clumping, like Alzheimer’s. These studies hold the potential to save the eyesight of millions of glaucoma patients.”
The findings have been the culmination of nearly two decades of research with Lieberman’s close collaborator, University of Texas at Austin chemical engineering Professor Jennifer Maynard, whose group helped discover the two antibodies that responded to the mutation. Lieberman’s group then worked to understand how the antibodies functioned, determining the two that most successfully broke down the protein.
“This study builds on 10 years of work that explains how myocilin folds to how to break it down,” Lieberman said. “I am at a very fortunate place in my career where this fundamental research coalesces into what we could use clinically.”
Treatment Transformation
Lieberman hopes the antibodies can help treat glaucoma patients, particularly those with early onset glaucoma, often children. She now has a research collaboration with Rebecca Neustein, a physician at Emory University who treats these young patients.
“She doesn't have much hope to give her patients for curing glaucoma,” Lieberman said. “So she was very excited that we could do some genotyping and figure out who these antibodies can help.”
Lieberman’s research offers a clearer future for millions suffering from glaucoma and those at risk of developing the disease. By leveraging antibodies to target and break down malfunctioning myocilin, this discovery not only paves the way for new treatments for glaucoma but also opens doors for addressing other protein-aggregation diseases like Alzheimer’s, Parkinson’s, and even Type 2 diabetes.
Funding: National Institutes of Health
Animation by Raul Perez
News Contact
Tess Malone, Senior Research Writer/Editor
tess.malone@gatech.edu
Pagination
- Previous page
- Page 11
- Next page






![<p><a href="https://doi.org/10.1643/CE-16-549">A series of experiment spanning 40 years on small islands in the Bahamas</a> have revealed how prey species, like small brown anole lizards (<em>Anolis sagrei</em>), <a href="https://doi.org/10.1038/nature03039">evolve in response to predators</a>, like the larger curly-tailed lizard (<em>Leiocepahlus carinatus</em>). Importantly, due to the long-term nature of this research, <a href="https://doi.org/10.1890/0012-9615(2002)072[0383:POACAL]2.0.CO;2">scientists were able to track ecosystem changes in response to this predator-driven rapid evolution</a>. (Illustration: Mark Belan/ArtSciStudios)</p> A series of experiment spanning 40 years on small islands in the Bahamas have revealed how prey species, like small brown anole lizards (Anolis sagrei), evolve in response to predators, like the larger curly-tailed lizard (Leiocepahlus carinatus). Importantly, due to the long-term nature of this research, scientists were able to track ecosystem changes in response to this predator-driven rapid evolution. (Illustration: Mark Belan/ArtSciStudios)](/sites/default/files/styles/wide/public/news/2026-05/JamesStroud_LizardImage.jpg.webp?itok=QhnA8bqJ)