Apr. 13, 2020
What-if questions can torment a doctor making coronavirus retest decisions: What if a patient’s initial negative test was a false negative, and he or she needs a second test? What if they don’t need it, and a retest would use up a scarce test kit and treatments that other patients need?
Such challenges led Piedmont Healthcare in Atlanta to establish a paper-based decision tree for ordering COVID-19 retests, and researchers at the Georgia Institute of Technology turned it into an automated digital tool. Piedmont further developed the tool and has now built it into the hospital’s electronic medical record, where it influences the ordering of retests.
A user can answer their “ifs” by clicking through questions, and the “if-this-then-do-that” algorithm makes recommendations for best courses of action, ranging from immediately treating a patient for COVID-19 to retesting to consulting a specialist. The final decision remains with the physician.
The questions are deceptively simple, but the recommendations are not always obvious. That reflects the algorithm’s usefulness to fill gaps in thinking about the new sickness, which can confront clinicians with surprises.
“If a patient has not had close contact with positive patients and the first test came back negative, a physician may think the patient does not need to be retested. But actually, the patient may need a second test because they are in intensive care and also have suspicious chest X-rays,” said Georgia Tech graduate research assistant April Yu, who converted the decision tree into a digital tool.
“One of our big worries in using a brand-new test like the coronavirus test is that it will miss real cases, and this tool helps prevent that,” said Dr. Bronwen Garner, who helped develop the original decision tree and is an infectious disease specialist at Piedmont Healthcare. “It also helps reassure physicians when they get a negative result that it is probably a true negative.”
Suspenseful decision-making
A physician’s reaction to an initial negative test can mean life or death because the physician not only decides on follow-up testing but also on treatment pathways and quarantine.
“If you make a misstep in the thought process, it can lead to cascading impacts not only for the patient but also for healthcare professionals and family members, who may be exposed to the patient,” said Pinar Keskinocak, William W. George Chair and Professor in Georgia Tech’s Stewart School of Industrial and Systems Engineering. “This tool is meant to help doctors easily stay on the decision tree path.”
Michael O’Toole, executive director of Piedmont Healthcare’s quality improvement department, originally pictured doctors getting an automated version of the decision tree to use on their phones. O’Toole called Keskinocak, and she tapped Yu, a member of her research group.
“Literally within four hours they had it ready for us. It was incredible,” said O’Toole, a Georgia Tech alumnus who studied industrial and systems engineering.
“It was a very pleasant surprise,” said Dr. Garner, who is also a Georgia Tech graduate. “Automated tools are better than a paper format because they’re in the same format as orders in our electronic system. We get notifications in real time instead of having to remember to check a piece of paper.”
The tool is in place in the system where doctors order retests and is specific to Piedmont’s workflow. It may not be directly transferable to other health care systems.
Piedmont Healthcare simplified the logic even more, and the hospital built its own custom alerts to guide physicians on retesting. For cases that are more ambiguous, Piedmont Healthcare’s final version of the tool also gives physicians inside the hospital guidance to consult with their in-house infectious disease specialists.
If-this-then-retest
In her original version, Yu had turned the decision tree criteria into a short panel of questions with yes and no answers. It took her six iterations to arrive at her final version.
Yu’s version asked whether the patient:
- has a relevant ailment
- previously tested positive for coronavirus
- is now in an intensive care unit
- has worsening lung conditions
- shows telltale lung damage in imaging
- has been diagnosed with a different ailment
- the patient has had contact with someone else who tested positive for coronavirus.
On the back end, the algorithm guided the user through risks of coronavirus presence based on the answers.
“The steps were easy to follow, and the answers were color-coded for urgency with white, yellow, and red,” said Keskinocak, who also directs Georgia Tech’s Center for Health and Humanitarian Systems.
One bright yellow answer read: “This patient needs re-testing 24 hours after the initial test!” And there were further recommendations on how to handle the case.
Here's how to subscribe to our free science and technology newsletter
Also read: Advice on DIY masks
Writer & Media Representative: Ben Brumfield (404-272-2780), email: ben.brumfield@comm.gatech.edu
Georgia Institute of Technology
Mar. 25, 2020
COVID-19 has caught Pinar Keskinocak well prepared. For years, she has studied how societies manage pandemics, and how outbreaks overtax the health care system and wrack supply chains to worsen pandemics. Here she shares her insights.
Empty classrooms and supermarket shelves marked the beginning of the COVID-19 pandemic. But Keskinocak expects more signs of the times to come – such as pop-up pandemic clinics and the shortage and rationing of medical supplies beyond masks and ventilators.
Keskinocak is the director of the Center for Health and Humanitarian Systems at the Georgia Institute of Technology, which studies how government and private sectors can cooperate to handle health and humanitarian crises. And she is William W. George Chair and Professor in Georgia Tech’s H. Milton Stewart School of Industrial and Systems Engineering.
In previous research, Keskinocak’s team created a model that accurately ran the course of the 1918 Spanish flu pandemic, and when COVID-19 struck, her team was already in the middle of modeling how special clinics could significantly slow a pandemic. In the meantime, temporary clinics in Wuhan, China, appear to have validated her model.
Healthcare expansion now
The surge of COVID-19 patients pushed Italy’s health care system into a very ugly crisis, and the U.S. needs to take measures now to handle similar patient surges. Pandemics often strike in two waves or more, and the second is usually the worst, so measures need to be lasting, Keskinocak said.
Even without COVID-19, the U.S. healthcare system has been under strain. Emergency rooms are often overcrowded; it takes a long time to schedule an appointment, and there is a chronic shortage of nursing staff.
[Read Keskinocak's guest op-ed in the New York Daily News: COVID clinics now]
“We need to expand capacity and unleash creative flexibility in our healthcare systems. We should use more telemedicine and create self-service stations for testing. I would particularly like to see specialized COVID-19 clinics established now,” Keskinocak said.
“Special clinics could be separate spaces in existing facilities or standalone facilities. As COVID-19 spreads, we expect a lot more people with cold- and flu-like symptoms to seek testing and care. The healthcare capacities are just not there for a business as usual approach, and taking it could harm patients by delaying care and increasing risk of infection.”
Gathering COVID-19 patients in tight spaces like waiting rooms with other patients would increase the coronavirus’ spread, and patients with preexisting conditions could face mortal threat. Contagion could also spread into hospitals.
“Dedicated pandemic clinics could implement targeted hygiene, air filtration, and specialized protective equipment beyond masks and gloves for healthcare workers. They can tailor workflows to test and care for patients quickly and effectively and keep them away from other patients and staff,” Keskinocak said.
Payment needs to be easy, too, including financing the uninsured. In the middle of a public health emergency, it is vital to not get bogged down by restrictions meant for normal times.
Potentially dangerous shortages
Toilet paper will make a comeback in supermarkets, but in its place, life-saving medications could become perilously scarce. Countries need to act now to prevent this from compounding the COVID-19 crisis.
“Dwindling availability of hospital beds, ventilators, and personal protective equipment like masks and gloves during a patient surge – those are the obvious things. But we could also see shortages of items like asthma medication or antidepressants. Worst case, even food supplies could run low,” Keskinocak said.
[Read Keskinocak's guest op-ed in The Hill: medical supply chain dangers]
Here’s how shortages work and can lead to price gouging and also rationing. The latter can have good effects.
“Shortages are the result of supply-demand imbalance caused by either an unexpected increase in demand or unexpected decrease in supply or both. Shortages are common in crises such as natural disasters or health emergencies. But given the worldwide slowdown of economic activity in pandemics, disruptions could get much worse this time,” Keskinocak said.
“Supply chains are actually intricate webs of multiple parts that span the globe. Pandemics damage many of those parts, and it can take time to recover. This creates a more serious and worrisome imbalance between supply and demand.”
Toilet paper will return because people fear-hoard it in a panic but consume it at normal rates. When the panic runs its course, demand slows back down to the actual rate of consumption and its normal supply chain, which is relatively simple, catches up.
“With medicine and healthcare services and supplies, the increase in demand is typically already in line with consumption, so a shortage in supply or increase in demand can create a supply-demand gap that continues for a long time,” Keskinocak said. “Medical supply chains are also very complex and fragile.”
Future vaccine distribution
In normal times, most supply chains work at a plodding pace, and when crisis strikes, it is tough to ramp them up due to expensive equipment, complex logistics, and strict regulations, particularly in health care. Even temporary shortages of medicines and medical devices can have consequences for patients who need them.
“If shortages become serious, rationing – with a priority allocation to those most in need – can help balance demand and supply for critical items like medications.”
Once created and approved, the production of vaccines or antivirals for COVID-19 will ramp up slowly and could be in short supply at first. Decision-makers need plan investments now in the supply chains necessary for their effective distribution.
This will include painful, necessary decisions like prioritizing first doses for healthcare workers, people with pre-existing conditions, and the elderly. The current system of restocking vaccines in the U.S. after initial distribution also has serious gaps that need fixing to save many more lives.
In the meantime, social distancing is one of the best ways to protect everyone and reduce the patient surge into clinics. Do it if you or anyone in your household has any cold-like symptoms.
[Read Keskinocak's commentary on social distancing on AJC.com]
Also read: Vaccine Supply Gaps Can Make Pandemics Deadlier
Media contacts: Ben Brumfield (ben.brumfield@comm.gatech.edu) and John Toon (john.toon@comm.gatech.edu)
Feb. 10, 2020
Four Georgia Institute of Technology faculty members have been elected as new members of the National Academy of Engineering (NAE). Marilyn Brown, Thomas Kurfess, Susan Margulies, and Alexander Shapiro join 83 other new NAE members for 2020 when they are formally inducted during a ceremony at the academy’s annual meeting on Oct. 4 in Washington, D.C.
Election of new NAE members, the culmination of a yearlong process, recognizes individuals who have made outstanding contributions to "engineering research, practice, or education, including, where appropriate, significant contributions to the engineering literature" and to "the pioneering of new and developing fields of technology, making major advancements in traditional fields of engineering, or developing/implementing innovative approaches to engineering education."
“It’s the honor of a lifetime to be recognized by the National Academy of Engineering for the impact we’ve have on understanding lung injuries in the critical care unit and traumatic brain injuries in children,” said Margulies, chair of the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University and, with Brown, one of just three women on the Georgia Tech faculty accorded NAE membership – one of the highest professional distinctions an engineer can receive.
“Our work is deeply collaborative, and I am grateful to the engineers, scientists, physicians, and patients who are partners in our journey,” Margulies added.
Margulies, a researcher in the Petit Institute for Bioengineering and Bioscience at Tech and a Georgia Research Alliance Eminent Scholar in Injury Biomechanics at Emory, was elected, “for elaborating the traumatic injury thresholds of brain and lung in terms of structure-function mechanisms,” according to the NAE announcement.
Using an integrated biomechanics approach, Margulies’ research program spans the micro-to-macro scales in two distinct areas, traumatic brain injury and ventilator-induced lung injury. Her work has generated new knowledge about the structural and functional responses of the brain and lungs to their mechanical environment. Margulies came to Georgia Tech in 2017 from the University of Pennsylvania, where she’d been a professor of bioengineering, and had earned her Master of Science in Engineering and Ph.D. in Bioengineering.
Brown, a Regents and Brook Byers Professor of Sustainable Systems in the School of Public Policy, was co-recipient of the Nobel Peace Prize in 2007 (for co-authorship of the Intergovernmental Panel on Climate Change Working Group III Assessment Report on Mitigation of Climate Change, Chapter 6).
She joined Georgia Tech in 2006 after a career at the U.S. Department of Energy's Oak Ridge National Laboratory, where she led several national climate change mitigation studies and became a leader in the analysis and interpretation of energy futures in the United States. Her research at Tech focuses on the design and impact of policies aimed at accelerating the development and deployment of sustainable energy technologies, emphasizing the electric utility industry. She was elected to NAE “for bridging engineering, social and behavioral sciences, and policy studies to achieve cleaner electric energy.”
Brown, who earned her Ph.D. at the Ohio State University, co-founded and chaired the Southeast Energy Efficiency Alliance, served two terms as a presidential appointee on the board of the Tennessee Valley Authority – the nation’s largest public power provider – and also served two terms on the U.S. Department of Energy’s Electricity Advisory Committee, where she led the Smart Grid Subcommittee.
“The most rewarding feature of my career has been working toward solutions with colleagues across disciplines,” Brown said.
Shapiro is the Russell Chandler III Chair and professor in the H. Milton Stewart School of Industrial and Systems Engineering, where his research is focused on stochastic programming, risk analysis, simulation-based optimization, and multivariate statistical analysis.
In 2013, he was awarded the INFORMS Khachiyan Prize for lifetime achievements in optimization. He received the 2018 Dantzig Prize from the Mathematical Optimization Society and the Society for Industrial and Applied Mathematics.
Since earning his Ph.D. in applied mathematics-statistics from Israel’s Ben-Gurion University of the Negev in 1981, Shapiro has made substantial contributions to the fields of optimization and large-scale, stochastic programming, and he was elected to NAE “for contributions to the theory, computation, and application of stochastic programming.”
Kurfess is professor and HUSCO/Ramirez Distinguished Chair in Fluid Power and Motion Control in the George W. Woodruff School of Mechanical Engineering, where he has helped guide the evolution of technology as a pioneer in the digital transformation of manufacturing.
Improving manufacturing technology is a pursuit that has roots in his childhood. “I grew up in my father’s machine shop,” said Kurfess, who has a special fondness for mom-and-pop operations. He was elected by the NAE “for development and implementation of innovative digital manufacturing technologies and system architectures.”
“I’m proud that the work we do has a positive impact on small and medium-sized enterprises, which are about 99% of the manufacturing operations, as well as large operations,” said Kurfess, who earned all of his degrees at MIT. “Our work targets people who are implementing the digital thread in manufacturing, and what the digital thread will do is make sure those smaller enterprises, those mom and pops, can have access to the latest and greatest technologies.”
Research News
Georgia Institute of Technology
177 North Avenue
Atlanta, Georgia 30332-0181 USA
Media Relations Contact: John Toon (404-894-6986) (jtoon@gatech.edu).
Writer: Jerry Grillo
News Contact
John Toon
Research News
(404) 894-6986
Feb. 10, 2020
Four Georgia Institute of Technology faculty members have been elected as new members of the National Academy of Engineering (NAE). Marilyn Brown, Thomas Kurfess, Susan Margulies, and Alexander Shapiro join 83 other new NAE members for 2020 when they are formally inducted during a ceremony at the academy’s annual meeting on Oct. 4 in Washington, D.C.
Election of new NAE members, the culmination of a yearlong process, recognizes individuals who have made outstanding contributions to "engineering research, practice, or education, including, where appropriate, significant contributions to the engineering literature" and to "the pioneering of new and developing fields of technology, making major advancements in traditional fields of engineering, or developing/implementing innovative approaches to engineering education."
“It’s the honor of a lifetime to be recognized by the National Academy of Engineering for the impact we’ve have on understanding lung injuries in the critical care unit and traumatic brain injuries in children,” said Margulies, chair of the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University and, with Brown and Elsa Reichmanis (elected in 1995), one of just three women on the Georgia Tech faculty accorded NAE membership – one of the highest professional distinctions an engineer can receive.
“Our work is deeply collaborative, and I am grateful to the engineers, scientists, physicians, and patients who are partners in our journey,” Margulies added.
Margulies, a researcher in the Petit Institute for Bioengineering and Bioscience at Tech and a Georgia Research Alliance Eminent Scholar in Injury Biomechanics at Emory, was elected, “for elaborating the traumatic injury thresholds of brain and lung in terms of structure-function mechanisms,” according to the NAE announcement.
Using an integrated biomechanics approach, Margulies’ research program spans the micro-to-macro scales in two distinct areas, traumatic brain injury and ventilator-induced lung injury. Her work has generated new knowledge about the structural and functional responses of the brain and lungs to their mechanical environment. Margulies came to Georgia Tech in 2017 from the University of Pennsylvania, where she’d been a professor of bioengineering, and had earned her Master of Science in Engineering and Ph.D. in Bioengineering.
Brown is Regents and Brook Byers Professor of Sustainable Systems in the School of Public Policy. Her deep expertise in climate and energy policy helped shape numerous reports for the Intergovernmental Panel on Climate Change, including one that led to the organization receiving the Nobel Peace Prize in 2007.
She joined Georgia Tech in 2006 after a career at the U.S. Department of Energy's Oak Ridge National Laboratory, where she led several national climate change mitigation studies and became a leader in the analysis and interpretation of energy futures in the United States. Her research at Tech focuses on the design and impact of policies aimed at accelerating the development and deployment of sustainable energy technologies, emphasizing the electric utility industry. She was elected to NAE “for bridging engineering, social and behavioral sciences, and policy studies to achieve cleaner electric energy.”
Brown, who earned her Ph.D. at the Ohio State University, co-founded and chaired the Southeast Energy Efficiency Alliance, served two terms as a presidential appointee on the board of the Tennessee Valley Authority – the nation’s largest public power provider – and also served two terms on the U.S. Department of Energy’s Electricity Advisory Committee, where she led the Smart Grid Subcommittee.
“The most rewarding feature of my career has been working toward solutions with colleagues across disciplines,” Brown said.
Shapiro is the Russell Chandler III Chair and professor in the H. Milton Stewart School of Industrial and Systems Engineering, where his research is focused on stochastic programming, risk analysis, simulation-based optimization, and multivariate statistical analysis.
In 2013, he was awarded the INFORMS Khachiyan Prize for lifetime achievements in optimization. He received the 2018 Dantzig Prize from the Mathematical Optimization Society and the Society for Industrial and Applied Mathematics.
Since earning his Ph.D. in applied mathematics-statistics from Israel’s Ben-Gurion University of the Negev in 1981, Shapiro has made substantial contributions to the fields of optimization and large-scale, stochastic programming, and he was elected to NAE “for contributions to the theory, computation, and application of stochastic programming.”
Kurfess is professor and HUSCO/Ramirez Distinguished Chair in Fluid Power and Motion Control in the George W. Woodruff School of Mechanical Engineering, where he has helped guide the evolution of technology as a pioneer in the digital transformation of manufacturing.
Improving manufacturing technology is a pursuit that has roots in his childhood. “I grew up in my father’s machine shop,” said Kurfess, who has a special fondness for mom-and-pop operations. He was elected by the NAE “for development and implementation of innovative digital manufacturing technologies and system architectures.”
“I’m proud that the work we do has a positive impact on small and medium-sized enterprises, which are about 99% of the manufacturing operations, as well as large operations,” said Kurfess, who earned all of his degrees at MIT. “Our work targets people who are implementing the digital thread in manufacturing, and what the digital thread will do is make sure those smaller enterprises, those mom and pops, can have access to the latest and greatest technologies.”
Research News
Georgia Institute of Technology
177 North Avenue
Atlanta, Georgia 30332-0181 USA
Media Relations Contact: John Toon (404-894-6986) (jtoon@gatech.edu).
Writer: Jerry Grillo
News Contact
John Toon
Research News
(404) 894-6986
Feb. 11, 2020
Four Georgia Institute of Technology faculty members have been elected as new members of the National Academy of Engineering (NAE). Marilyn Brown, Thomas Kurfess, Susan Margulies, and Alexander Shapiro join 83 other new NAE members for 2020 when they are formally inducted during a ceremony at the academy’s annual meeting on Oct. 4 in Washington, D.C.
Election of new NAE members, the culmination of a yearlong process, recognizes individuals who have made outstanding contributions to "engineering research, practice, or education, including, where appropriate, significant contributions to the engineering literature" and to "the pioneering of new and developing fields of technology, making major advancements in traditional fields of engineering, or developing/implementing innovative approaches to engineering education."
“It’s the honor of a lifetime to be recognized by the National Academy of Engineering for the impact we’ve have on understanding lung injuries in the critical care unit and traumatic brain injuries in children,” said Margulies, chair of the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University and, with Brown, one of just three women on the Georgia Tech faculty accorded NAE membership – one of the highest professional distinctions an engineer can receive.
“Our work is deeply collaborative, and I am grateful to the engineers, scientists, physicians, and patients who are partners in our journey,” Margulies added.
Margulies, a researcher in the Petit Institute for Bioengineering and Bioscience at Tech and a Georgia Research Alliance Eminent Scholar in Injury Biomechanics at Emory, was elected, “for elaborating the traumatic injury thresholds of brain and lung in terms of structure-function mechanisms,” according to the NAE announcement.
Using an integrated biomechanics approach, Margulies’ research program spans the micro-to-macro scales in two distinct areas, traumatic brain injury and ventilator-induced lung injury. Her work has generated new knowledge about the structural and functional responses of the brain and lungs to their mechanical environment. Margulies came to Georgia Tech in 2017 from the University of Pennsylvania, where she’d been a professor of bioengineering, and had earned her Master of Science in Engineering and Ph.D. in Bioengineering.
Brown, a Regents and Brook Byers Professor of Sustainable Systems in the School of Public Policy, was co-recipient of the Nobel Peace Prize in 2007 (for co-authorship of the Intergovernmental Panel on Climate Change Working Group III Assessment Report on Mitigation of Climate Change, Chapter 6).
She joined Georgia Tech in 2006 after a career at the U.S. Department of Energy's Oak Ridge National Laboratory, where she led several national climate change mitigation studies and became a leader in the analysis and interpretation of energy futures in the United States. Her research at Tech focuses on the design and impact of policies aimed at accelerating the development and deployment of sustainable energy technologies, emphasizing the electric utility industry. She was elected to NAE “for bridging engineering, social and behavioral sciences, and policy studies to achieve cleaner electric energy.”
Brown, who earned her Ph.D. at the Ohio State University, co-founded and chaired the Southeast Energy Efficiency Alliance, served two terms as a presidential appointee on the board of the Tennessee Valley Authority – the nation’s largest public power provider – and also served two terms on the U.S. Department of Energy’s Electricity Advisory Committee, where she led the Smart Grid Subcommittee.
“The most rewarding feature of my career has been working toward solutions with colleagues across disciplines,” Brown said.
Shapiro is the Russell Chandler III Chair and professor in the H. Milton Stewart School of Industrial and Systems Engineering, where his research is focused on stochastic programming, risk analysis, simulation-based optimization, and multivariate statistical analysis.
In 2013, he was awarded the INFORMS Khachiyan Prize for lifetime achievements in optimization. He received the 2018 Dantzig Prize from the Mathematical Optimization Society and the Society for Industrial and Applied Mathematics.
Since earning his Ph.D. in applied mathematics-statistics from Israel’s Ben-Gurion University of the Negev in 1981, Shapiro has made substantial contributions to the fields of optimization and large-scale, stochastic programming, and he was elected to NAE “for contributions to the theory, computation, and application of stochastic programming.”
Kurfess is professor and HUSCO/Ramirez Distinguished Chair in Fluid Power and Motion Control in the George W. Woodruff School of Mechanical Engineering, where he has helped guide the evolution of technology as a pioneer in the digital transformation of manufacturing.
Improving manufacturing technology is a pursuit that has roots in his childhood. “I grew up in my father’s machine shop,” said Kurfess, who has a special fondness for mom-and-pop operations. He was elected by the NAE “for development and implementation of innovative digital manufacturing technologies and system architectures.”
“I’m proud that the work we do has a positive impact on small and medium-sized enterprises, which are about 99% of the manufacturing operations, as well as large operations,” said Kurfess, who earned all of his degrees at MIT. “Our work targets people who are implementing the digital thread in manufacturing, and what the digital thread will do is make sure those smaller enterprises, those mom and pops, can have access to the latest and greatest technologies.”
Research News
Georgia Institute of Technology
177 North Avenue
Atlanta, Georgia 30332-0181 USA
Media Relations Contact: John Toon (404-894-6986) (jtoon@gatech.edu).
Writer: Jerry Grillo
News Contact
John Toon
Research News
(404) 894-6986
May. 10, 2012
New research findings show that embryonic stem cells unable to fully compact the DNA inside them cannot complete their primary task: differentiation into specific cell types that give rise to the various types of tissues and structures in the body.
Researchers from the Georgia Institute of Technology and Emory University found that chromatin compaction is required for proper embryonic stem cell differentiation to occur. Chromatin, which is composed of histone proteins and DNA, packages DNA into a smaller volume so that it fits inside a cell.
A study published on May 10, 2012 in the journal PLoS Genetics found that embryonic stem cells lacking several histone H1 subtypes and exhibiting reduced chromatin compaction suffered from impaired differentiation under multiple scenarios and demonstrated inefficiency in silencing genes that must be suppressed to induce differentiation.
“While researchers have observed that embryonic stem cells exhibit a relaxed, open chromatin structure and differentiated cells exhibit a compact chromatin structure, our study is the first to show that this compaction is not a mere consequence of the differentiation process but is instead a necessity for differentiation to proceed normally,” said Yuhong Fan, an assistant professor in the Georgia Tech School of Biology.
Fan and Todd McDevitt, an associate professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University, led the study with assistance from Georgia Tech graduate students Yunzhe Zhang and Kaixiang Cao, research technician Marissa Cooke, and postdoctoral fellow Shiraj Panjwani.
The work was supported by the National Institutes of Health’s National Institute of General Medical Sciences (NIGMS), the National Science Foundation, a Georgia Cancer Coalition Distinguished Scholar Award, and a Johnson & Johnson/Georgia Tech Healthcare Innovation Award.
To investigate the impact of linker histones and chromatin folding on stem cell differentiation, the researchers used embryonic stem cells that lacked three subtypes of linker histone H1 -- H1c, H1d and H1e -- which is the structural protein that facilitates the folding of chromatin into a higher-order structure. They found that the expression levels of these H1 subtypes increased during embryonic stem cell differentiation, and embryonic stem cells lacking these H1s resisted spontaneous differentiation for a prolonged time, showed impairment during embryoid body differentiation and were unsuccessful in forming a high-quality network of neural cells.
“This study has uncovered a new, regulatory function for histone H1, a protein known mostly for its role as a structural component of chromosomes,” said Anthony Carter, who oversees epigenetics grants at NIGMS. “By showing that H1 plays a part in controlling genes that direct embryonic stem cell differentiation, the study expands our understanding of H1’s function and offers valuable new insights into the cellular processes that induce stem cells to change into specific cell types.”
During spontaneous differentiation, the majority of the H1 triple-knockout embryonic stem cells studied by the researchers retained a tightly packed colony structure typical of undifferentiated cells and expressed high levels of Oct4 for a prolonged time. Oct4 is a pluripotency gene that maintains an embryonic stem cell’s ability to self-renew and must be suppressed to induce differentiation.
“H1 depletion impaired the suppression of the Oct4 and Nanog pluripotency genes, suggesting a novel mechanistic link by which H1 and chromatin compaction may mediate pluripotent stem cell differentiation by contributing to the epigenetic silencing of pluripotency genes,” explained Fan. “While a significant reduction in H1 levels does not interfere with embryonic stem cell self-renewal, it appears to impair differentiation.”
The researchers also used a rotary suspension culture method developed by McDevitt to produce with high efficiency homogonous 3D clumps of embryonic stem cells called embryoid bodies. Embryoid bodies typically contain cell types from all three germ layers -- the ectoderm, mesoderm and endoderm -- that give rise to the various types of tissues and structures in the body. However, the majority of the H1 triple-knockout embryoid bodies formed in rotary suspension culture lacked differentiated structures and displayed gene expression signatures characteristic of undifferentiated stem cells.
“H1 triple-knockout embryoid bodies displayed a reduced level of activation of many developmental genes and markers in rotary culture, suggesting that differentiation to all three germ layers was affected.” noted McDevitt.
The embryoid bodies also lacked the epigentic changes at the pluripotency genes necessary for differentiation, according to Fan.
“When we added one of the deleted H1 subtypes to the embryoid bodies, Oct4 was suppressed normally and embryoid body differentiation continued,” explained Fan. “The epigenetic regulation of Oct4 expression by H1 was also evident in mouse embryos.”
In another experiment, the researchers provided an environment that would encourage embryonic stem cells to differentiate into neural cells. However, the H1 triple-knockout cells were defective in forming neuronal and glial cells and a neural network, which is essential for nervous system development. Only 10 percent of the H1 triple-knockout embryoid bodies formed neurites and they produced on average eight neurites each. In contrast, half of the normal embryoid bodies produced, on average, 18 neurites.
In future work, the researchers plan to investigate whether controlling H1 histone levels can be used to influence the reprogramming of adult cells to obtain induced pluripotent stem cells, which are capable of differentiating into tissues in a way similar to embryonic stem cells.
Research reported in this publication was supported by the National Institute of General Medical Sciences of the National Institutes of Health (NIH) under award number GM085261 and the National Science Foundation under award number CBET-0939511. The content is solely the responsibility of the principal investigators and does not necessarily represent the official views of the NIH or NSF.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
abby@innovate.gatech.edu
404-385-3364
May. 06, 2012
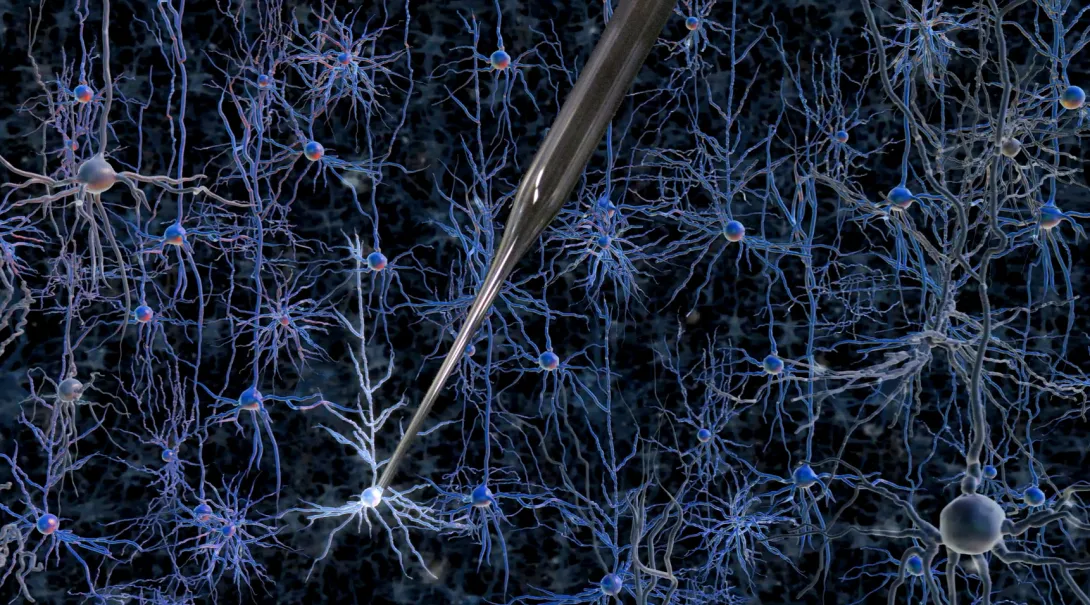
Gaining access to the inner workings of a neuron in the living brain offers a wealth of useful information: its patterns of electrical activity, its shape, even a profile of which genes are turned on at a given moment. However, achieving this entry is such a painstaking task that it is considered an art form; it is so difficult to learn that only a small number of labs in the world practice it.
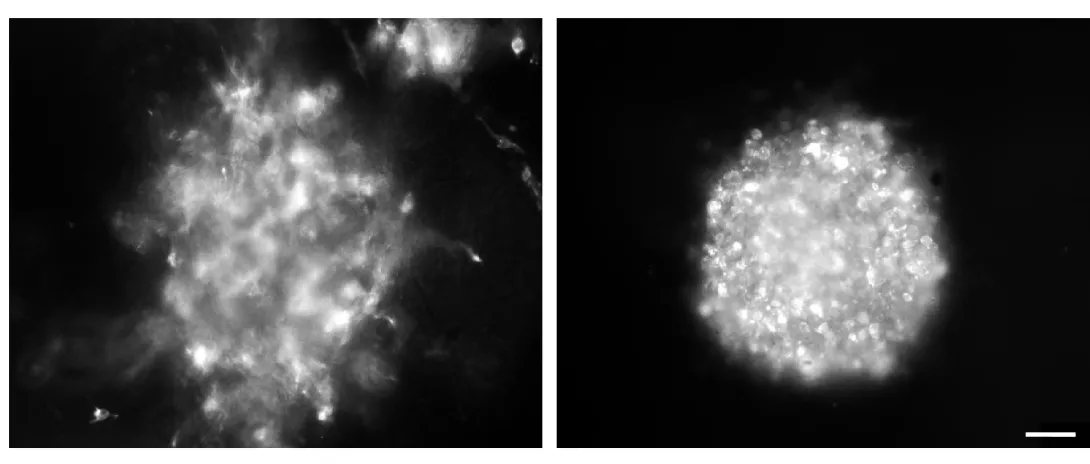
But that could soon change: Researchers at MIT and the Georgia Institute of Technology have developed a way to automate the process of finding and recording information from neurons in the living brain. The researchers have shown that a robotic arm guided by a cell-detecting computer algorithm can identify and record from neurons in the living mouse brain with better accuracy and speed than a human experimenter.
The new automated process eliminates the need for months of training and provides long-sought information about living cells’ activities. Using this technique, scientists could classify the thousands of different types of cells in the brain, map how they connect to each other, and figure out how diseased cells differ from normal cells.
The project is a collaboration between the labs of Ed Boyden, associate professor of biological engineering and brain and cognitive sciences at MIT, and Craig Forest, an assistant professor in the George W. Woodruff School of Mechanical Engineering at Georgia Tech.
“Our team has been interdisciplinary from the beginning, and this has enabled us to bring the principles of precision machine design to bear upon the study of the living brain,” Forest says. His graduate student, Suhasa Kodandaramaiah, spent the past two years as a visiting student at MIT, and is the lead author of the study, which appears in the May 6 issue of Nature Methods.
The method could be particularly useful in studying brain disorders such as schizophrenia, Parkinson’s disease, autism and epilepsy, Boyden says. “In all these cases, a molecular description of a cell that is integrated with [its] electrical and circuit properties … has remained elusive,” says Boyden, who is a member of MIT’s Media Lab and McGovern Institute for Brain Research. “If we could really describe how diseases change molecules in specific cells within the living brain, it might enable better drug targets to be found.”
Automation
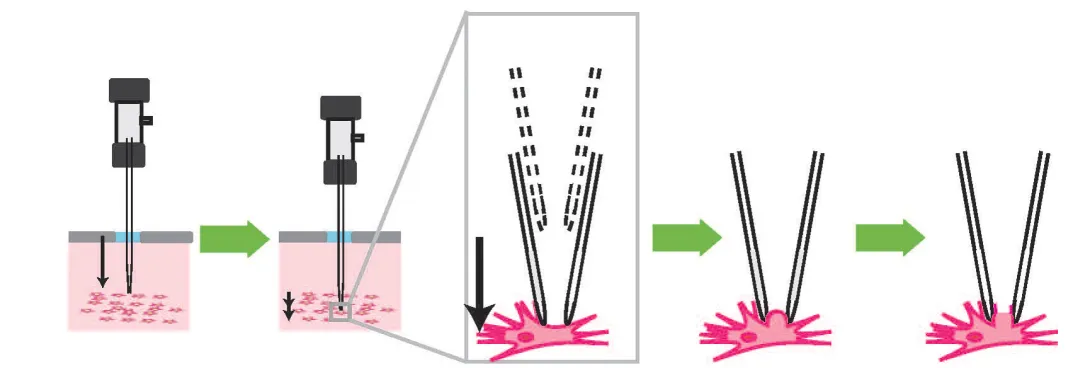
Kodandaramaiah, Boyden and Forest set out to automate a 30-year-old technique known as whole-cell patch clamping, which involves bringing a tiny hollow glass pipette in contact with the cell membrane of a neuron, then opening up a small pore in the membrane to record the electrical activity within the cell. This skill usually takes a graduate student or postdoc several months to learn.
Kodandaramaiah spent about four months learning the manual patch-clamp technique, giving him an appreciation for its difficulty. “When I got reasonably good at it, I could sense that even though it is an art form, it can be reduced to a set of stereotyped tasks and decisions that could be executed by a robot,” he says.
To that end, Kodandaramaiah and his colleagues built a robotic arm that lowers a glass pipette into the brain of an anesthetized mouse with micrometer accuracy. As it moves, the pipette monitors a property called electrical impedance — a measure of how difficult it is for electricity to flow out of the pipette. If there are no cells around, electricity flows and impedance is low. When the tip hits a cell, electricity can’t flow as well and impedance goes up.
The pipette takes two-micrometer steps, measuring impedance 10 times per second. Once it detects a cell, it can stop instantly, preventing it from poking through the membrane. “This is something a robot can do that a human can’t,” Boyden says.
Once the pipette finds a cell, it applies suction to form a seal with the cell’s membrane. Then, the electrode can break through the membrane to record the cell’s internal electrical activity. The robotic system can detect cells with 90 percent accuracy, and establish a connection with the detected cells about 40 percent of the time.
The researchers also showed that their method can be used to determine the shape of the cell by injecting a dye; they are now working on extracting a cell’s contents to read its genetic profile.
Development of the new technology was funded primarily by the National Institutes of Health, the National Science Foundation and the MIT Media Lab.
New era for robotics
The researchers recently created a startup company, Neuromatic Devices, to commercialize the device.
The researchers are now working on scaling up the number of electrodes so they can record from multiple neurons at a time, potentially allowing them to determine how different parts of the brain are connected.
They are also working with collaborators to start classifying the thousands of types of neurons found in the brain. This “parts list” for the brain would identify neurons not only by their shape — which is the most common means of classification — but also by their electrical activity and genetic profile.
“If you really want to know what a neuron is, you can look at the shape, and you can look at how it fires. Then, if you pull out the genetic information, you can really know what’s going on,” Forest says. “Now you know everything. That’s the whole picture.”
Boyden says he believes this is just the beginning of using robotics in neuroscience to study living animals. A robot like this could potentially be used to infuse drugs at targeted points in the brain, or to deliver gene therapy vectors. He hopes it will also inspire neuroscientists to pursue other kinds of robotic automation — such as in optogenetics, the use of light to perturb targeted neural circuits and determine the causal role that neurons play in brain functions.
Neuroscience is one of the few areas of biology in which robots have yet to make a big impact, Boyden says. “The genome project was done by humans and a giant set of robots that would do all the genome sequencing. In directed evolution or in synthetic biology, robots do a lot of the molecular biology,” he says. “In other parts of biology, robots are essential.”
Other co-authors include MIT grad student Giovanni Talei Franzesi and MIT postdoc Brian Y. Chow.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or Caroline McCall (cmccall5@mit.edu; 617-253-1682)
Writer: Anne Trafton, MIT News
News Contact
Abby Robinson
Research News and Publications
abby@innovate.gatech.edu
404-385-3364
Apr. 24, 2012
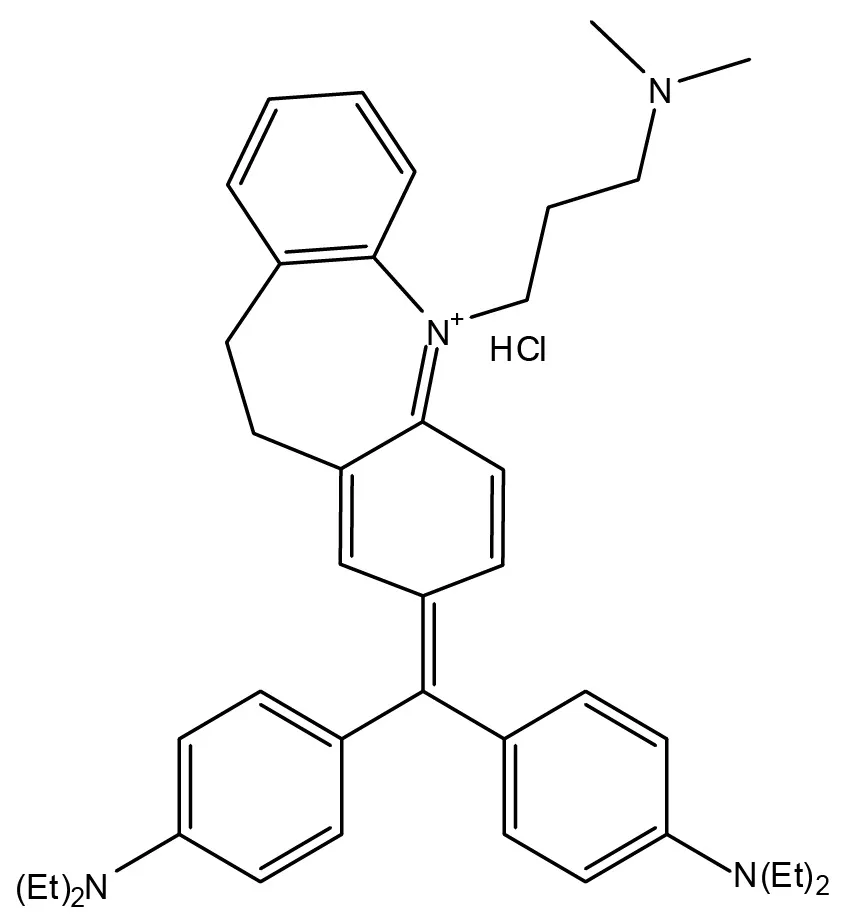
Fibronectin plays a major role in wound healing and embryonic development. The protein, which is located in the extracellular matrix of cells, has also been linked to pathological conditions including cancer and fibrosis.
During physiological processes, fibronectin fibers are believed to experience mechanical forces that strain the fibers and cause dramatic structural modifications that change their biological activity. While understanding the role of fibronectin strain events in development and disease progression is becoming increasingly important, detecting and interrogating these events is difficult.
In a new study, researchers identified molecular probes capable of selectively attaching to fibronectin fibers under different strain states, enabling the detection and examination of fibronectin strain events in both culture and living tissues.
“The mechano-sensitive molecular probes we identified allow us to dynamically examine the relevance of mechanical strain events within the natural cellular microenvironment and correlate these events with specific alterations in fibronectin associated with the progression of disease,” said Thomas Barker, an assistant professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
The study was published on April 23, 2012 in the online early edition of the journal Proceedings of the National Academy of Sciences. Barker worked on the study with Georgia Tech graduate student Lizhi Cao and Harry Bermudez, an assistant professor in the University of Massachusetts Amherst Department of Polymer Science and Engineering. The research was supported by the National Institutes of Health.
Researchers have hypothesized that mechanical forces emanating from cells may partially unfold fibronectin and regulate what proteins bind to it. While simulation and tissue culture experiments support this hypothesis, direct evidence that such molecular events occur in living organisms has not yet been presented, according to Barker.
A technique called intramolecular fluorescence resonance energy transfer (FRET) has been used to detect molecular strain events in fibronectin fibers, but the technique has limitations because it cannot be used on living tissues and requires the fibronectin to be chemically labeled.
“The molecular probes we identified can be used to map molecular strain events in native extracellular matrix and living lung tissues,” explained Barker. “The probes can also be used to study the mechanism by which cells control the mechanical forces that alter fibronectin’s conformation, control the exposure of its binding sites and regulate cell signaling.”
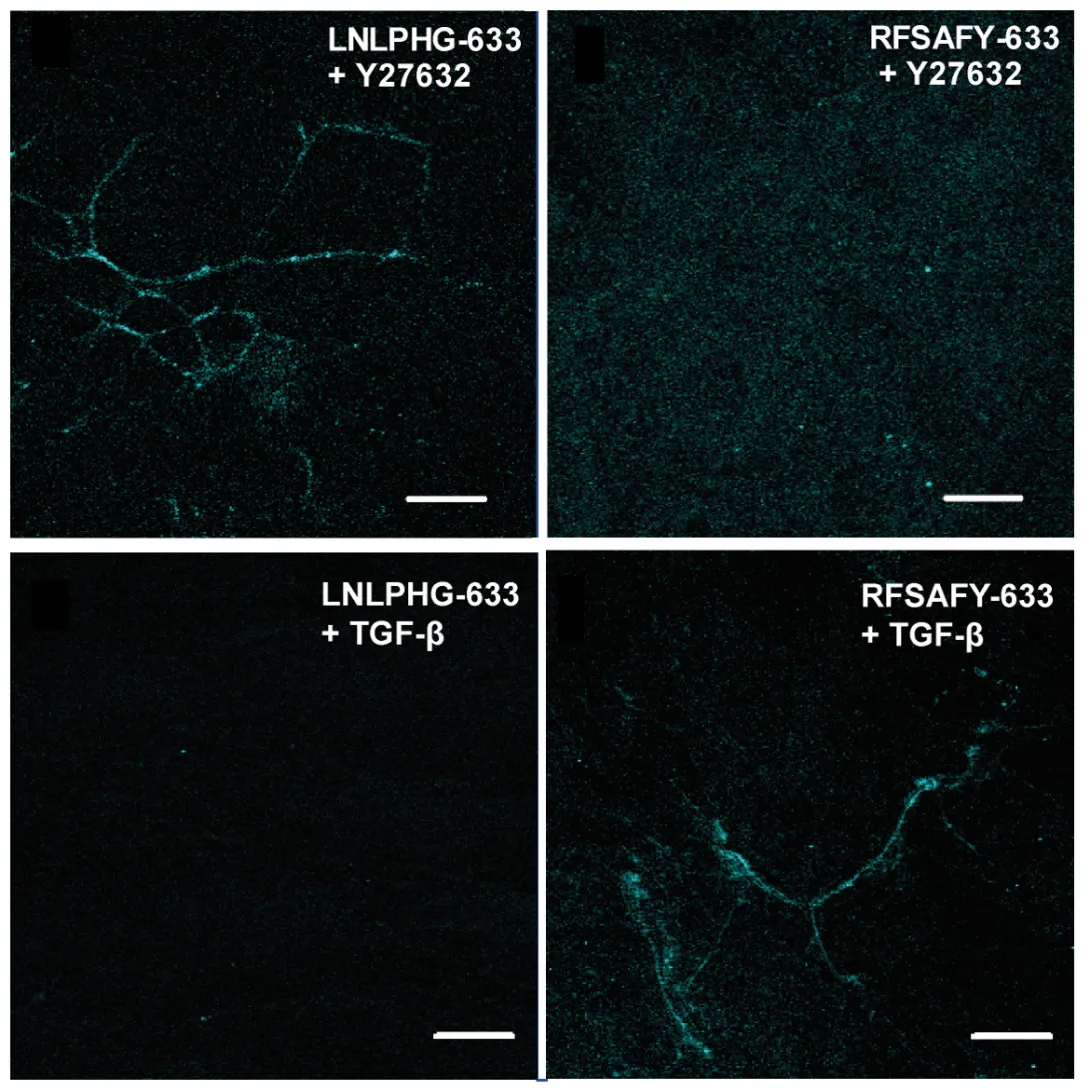
The researchers used a controlled fibronectin fiber deposition and extension technique to apply tension to the fibers and stretch them to 2.6 times their original length without significant breakage. Then they used a technique called phage display to identify peptides capable of discriminating fibronectin fibers under relaxed and strained conditions. The molecular probes displaying peptide sequences LNLPHG and RFSAFY showed the greatest binding affinity to fibronectin fibers and the greatest efficiency in discriminating between relaxed and strained fibers.
For proof-of-concept demonstrations, the researchers used the probes to discriminate fibronectin fibers within native extracellular matrix and mouse lung slices. LNLPHG preferentially attached to relaxed fibronectin fibers, whereas RFSAFY bound to strained fibers. The probes never attached to the same fiber, which confirmed their ability to selectively discriminate regions within a fibronectin fiber network.
“This study strongly suggests that fibronectin fibers under strain display markedly different biochemical signatures that can be used for the molecular-level detection of fibronectin fiber strain,” explained Barker. “The data also show the potential for living tissue to be interrogated for mechano-chemical alterations that lead to physiological and pathological progression.”
In the future, the researchers hope to use these fibronectin strain-sensitive probes to target therapeutics to fibronectin fibers based on their mechanical signature.
This work was supported in part by training grants from the National Institutes of Health (NIH) (Award Nos. T32-GM008433 and T32-EB006343). The content is solely the responsibility of the principal investigators and does not necessarily represent the official views of the NIH.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
abby@innovate.gatech.edu
404-385-3364
Mar. 28, 2012
Treating invasive brain tumors with a combination of chemotherapy and radiation has improved clinical outcomes, but few patients survive longer than two years after diagnosis. The effectiveness of the treatment is limited by the tumor’s aggressive invasion of healthy brain tissue, which restricts chemotherapy access to the cancer cells and complicates surgical removal of the tumor.
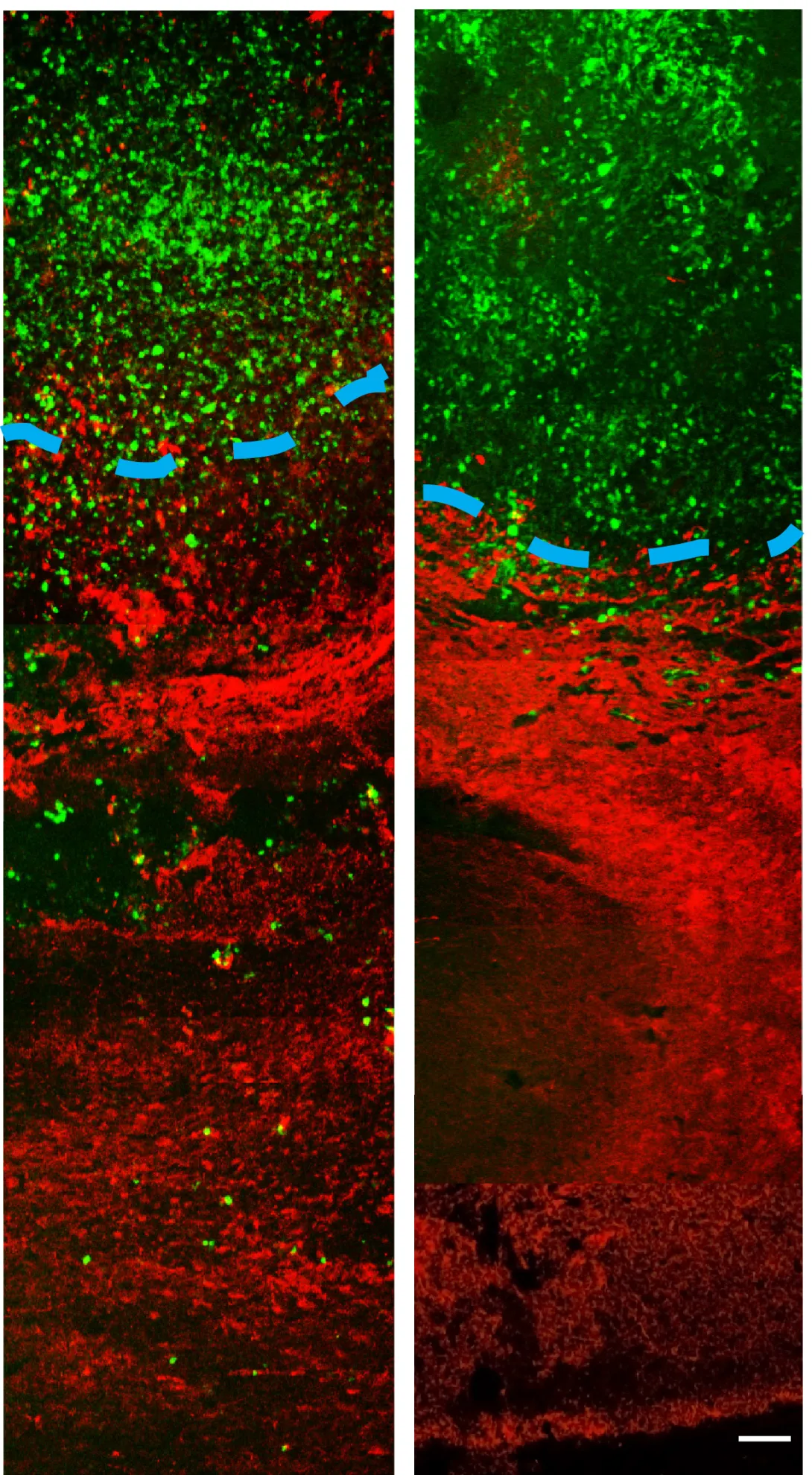
To address this challenge, researchers from the Georgia Institute of Technology and Emory University have designed a new treatment approach that appears to halt the spread of cancer cells into normal brain tissue in animal models. The researchers treated animals possessing an invasive tumor with a vesicle carrying a molecule called imipramine blue, followed by conventional doxorubicin chemotherapy. The tumors ceased their invasion of healthy tissue and the animals survived longer than animals treated with chemotherapy alone.
“Our results show that imipramine blue stops tumor invasion into healthy tissue and enhances the efficacy of chemotherapy, which suggests that chemotherapy may be more effective when the target is stationary,” said Ravi Bellamkonda, a professor in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University. “These results reveal a new strategy for treating brain cancer that could improve clinical outcomes.”
The results of this work were published on March 28, 2012 in the journal Science Translational Medicine. The research was supported primarily by the Ian’s Friends Foundation and partially by the Georgia Cancer Coalition, the Wallace H. Coulter Foundation and a National Science Foundation graduate research fellowship.
In addition to Bellamkonda, collaborators on the project include Jack Arbiser, a professor in the Emory University Department of Dermatology; Daniel Brat, a professor in the Emory University Department of Pathology and Laboratory Medicine; and the paper’s lead author, Jennifer Munson, a former Fulbright Scholar who was a bioengineering graduate student in the Georgia Tech School of Chemical & Biomolecular Engineering when the research was conducted.
Arbiser designed the novel imipramine blue compound, which is an organic triphenylmethane dye. After in vitro experiments showed that imipramine blue effectively inhibited movement of several cancer cell lines, the researchers tested the compound in an animal model of aggressive cancer that exhibited attributes similar to a human brain tumor called glioblastoma.
“There were many reasons why we chose to use the RT2 astrocytoma rat model for these experiments,” said Brat. “The tumor exhibited properties of aggressive growth, invasiveness, angiogenesis and necrosis that are similar to human glioblastoma; the model utilized an intact immune system, which is seen in the human disease; and the model enabled increased visualization by MRI because it was a rat model, rather than a mouse.”
Because imipramine blue is hydrophobic and doxorubicin is cytotoxic, the researchers encapsulated each compound in an artificially-prepared vesicle called a liposome so that the drugs would reach the brain. The liposomal drug delivery vehicle also ensured that the drugs would not be released into tissue until they passed through leaky blood vessel walls, which are only present where a tumor is growing.
Animals received one of the following four treatments: liposomes filled with saline, liposomes filled with imipramine blue, liposomes filled with doxorubicin chemotherapy, or liposomes filled with imipramine blue followed by liposomes filled with doxorubicin chemotherapy.
All of the animals that received the sequential treatment of imipramine blue followed by doxorubicin chemotherapy survived for 200 days -- more than 6 months -- with no observable tumor mass. Of the animals treated with doxorubicin chemotherapy alone, 33 percent were alive after 200 days with a median survival time of 44 days. Animals that received capsules filled with saline or imipramine blue – but no chemotherapy -- did not survive more than 19 days.
“Our results show that the increased effectiveness of the chemotherapy treatment is not because of a synergistic toxicity between imipramine blue and doxorubicin. Imipramine blue is not making the doxorubicin more toxic, it’s simply stopping the movement of the cancer cells and containing the cancer so that the chemotherapy can do a better job,” explained Bellamkonda, who is also the Carol Ann and David D. Flanagan Chair in Biomedical Engineering and a Georgia Cancer Coalition Distinguished Cancer Scholar.
MRI results showed a reduction and compaction of the tumor in animals treated with imipramine blue followed by doxorubicin chemotherapy, while animals treated with chemotherapy alone presented with abnormal tissue and glioma cells. MRI also indicated that the blood-brain barrier breach often seen during tumor growth was present in the animals treated with chemotherapy alone, but not the group treated with chemotherapy and imipramine blue.
According to the researchers, imipramine blue appears to improve the outcome of brain cancer treatment by altering the regulation of actin, a protein found in all eukaryotic cells. Actin mediates a variety of essential biological functions, including the production of reactive oxygen species. Most cancer cells exhibit overproduction of reactive oxygen species, which are thought to stimulate cancer cells to invade healthy tissue. The dye’s reorganization of the actin cytoskeleton is thought to inhibit production of enzymes that produce reactive oxygen species.
“I formulated the imipramine blue compound as a triphenylmethane dye because I knew that another triphenylmethane dye, gentian violet, exhibited anti-cancer properties, and I decided to use imipramine -- a drug used to treat depression -- as the starting material because I knew it could get into the brain,” said Arbiser.
For future studies, the researchers are planning to test imipramine blue’s effect on animal models with invasive brain tumors, metastatic tumors, and other types of cancer such as prostate and breast.
“While we need to conduct future studies to determine if the effect of imipramine blue is the same for different types of cancer diagnosed at different stages, this initial study shows the possibility that imipramine blue may be useful as soon as any tumor is diagnosed, before anti-cancer treatment begins, to create a more treatable tumor and enhance clinical outcome,” noted Bellamkonda.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
abby@innovate.gatech.edu
404-385-3364
Feb. 13, 2012
Valentine’s Day evokes images of a stylized heart shape, but for a group of Georgia Institute of Technology researchers, the heart is a complex organ that interests them throughout the year.
Georgia Tech researchers are developing new ways to diagnose and treat heart problems -- from advanced imaging techniques and guidance for drug therapies to sophisticated surgical procedures. Georgia Tech’s emphasis on translational research accelerates the pace at which new heart-related discoveries are put to use in patient care.
Improving Heart Surgery
To advance the goal of minimally invasive cardiac surgery, researchers have developed a technology that simplifies and standardizes the technique for opening and closing the beating heart during surgery.
Apica Cardiovascular, a Georgia Tech and Emory University medical device startup, licensed the technology from the two institutions. The firm recently received a $5.5 million investment to further develop the system, which will make the transapical access and closure procedure required for delivering therapeutic devices to the heart more routine for cardiac surgeons. The goal is to expand the use of surgery techniques that are less invasive and do not require stopping the heart.
With research and development support from the Coulter Foundation Translational Research Program and the Georgia Research Alliance, the company has already completed a series of pre-clinical studies to test the functionality of the device and its biocompatibility. James Greene currently serves as the CEO of the company, which has offices in Galway, Ireland, and in Atlanta.
For more information on this work, visit http://gtresearchnews.gatech.edu/apica-cardiovascular/.
Diagnosing Heart Disease
Levent Degertekin is designing tiny devices micromachined from silicon that may make diagnosing and treating coronary artery diseases easier.
Degertekin, the George W. Woodruff Chair in Mechanical Systems, and Paul Hasler, a professor in the School of Electrical and Computer Engineering at Georgia Tech, micromachined intravascular ultrasound imaging arrays with integrated electronics. Placed on catheters inserted into the body, the devices image the arteries of the heart in three dimensions at high resolution using high-frequency ultrasound waves.
The system boasts a more compact design and three-dimensional imaging capability for guiding cardiologists during interventions, such as those for completely blocked arteries. The technology also offers higher resolution than current intravascular ultrasound systems, which help diagnose vulnerable plaque, a leading cause of heart attacks.
Funding for this research currently is provided by the National Institutes of Health. To commercialize the technology, the researchers have formed a startup company called SIBUS Medical, which is receiving assistance from VentureLab, a unit of Georgia Tech’s Enterprise Innovation Institute that nurtures faculty startup companies.
Detecting and Treating Atherosclerosis
With a five-year $14.6 million contract from the National Institutes of Health (NIH), Georgia Tech and Emory University researchers are developing nanotechnology and biomolecular engineering tools and methodologies for detecting and treating atherosclerosis. The award supports the interdisciplinary Center for Translational Cardiovascular Nanomedicine, which is led by Gang Bao, the Robert A. Milton Chair in Biomedical Engineering in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
Atherosclerosis typically occurs in branched or curved regions of arteries where plaques form because of cholesterol build-up. Inflammation can alter the structure of plaques so they become more likely to rupture, potentially causing a blood vessel blockage and leading to heart attack or stroke.
The researchers are working to accomplish four goals:
- Using nanoparticle probes to image and characterize atherosclerotic plaques
- Diagnosing cardiovascular disease from a blood sample
- Designing new methods for delivering anti-atherosclerosis drugs and genes into the body
- Developing stem cell based therapies to repair damaged heart tissue
Additional researchers from the Coulter Department and from Emory University are also contributing to the project. For more information on this work, visit http://gtresearchnews.gatech.edu/cardiovascular-nanomedicine-center/.
Improving Drug Dosing Following a Heart Attack
A research team led by Georgia Tech mechanical engineering assistant professor Craig Forest is designing a device to quickly and accurately personalize a patient’s drug dosage to prevent blood clots that can cause heart attacks.
When someone experiencing heart attack symptoms arrives at an emergency room, he or she typically receives a standard dose of aspirin and/or clopidogrel to prevent further blood clotting. But that standard dose may not be the best dose for a given individual.
With Forest’s device, a small blood sample is sent through a microchip containing a network of microfabricated capillaries that mimic the branching coronary arteries around the human heart. Because the branches contain flow restrictions of different sizes, the failure of blood to flow through the branches with smaller restrictions indicates that a higher drug dose may be required.
Determining the necessary dose of anti-clotting drugs can be difficult. Too much of the drug may cause the patient to experience gastrointestinal bleeding. Too little drug may allow additional clot formation and set the stage for another heart attack. Forest’s device should help determine the right dosage for each patient.
Emory University Department of Emergency Medicine assistant professor Jeremy Ackerman and Georgia Tech Regents’ professor of mechanical engineering David Ku are working with Forest on this project, which is supported by the American Heart Association.
Examining Heart Valve Leakage
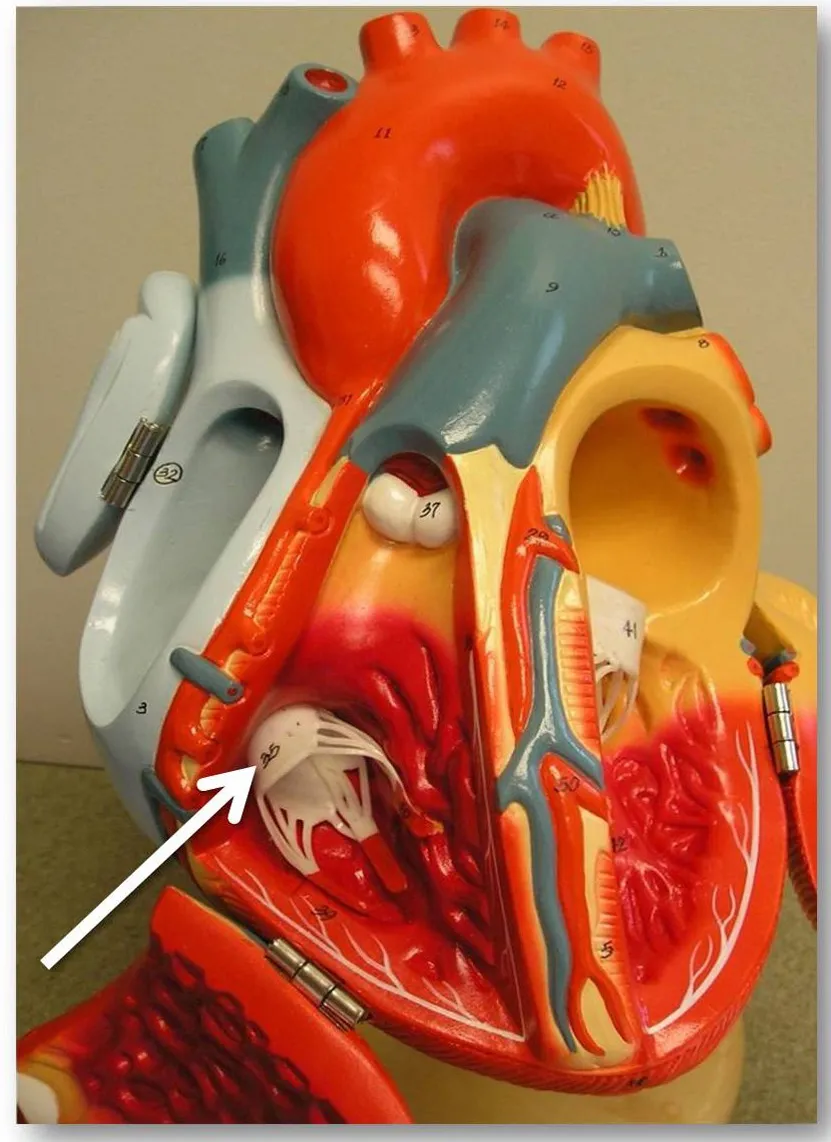
An estimated 1.6 million Americans suffer moderate to severe leakage through their tricuspid valve, a complex structure that closes off the heart’s right ventricle from the right atrium. If left untreated, severe leakage can affect an individual’s quality of life and can even lead to death.
Research teams led by Ajit Yoganathan, Georgia Tech Regents’ professor and Wallace H. Coulter Distinguished Faculty Chair in Biomedical Engineering, have discovered causes for the tricuspid valve’s leakage and ways to predict the severity of leakage in the valve. These study results could lead to improved diagnosis and treatment of the condition.
A study published in the journal Circulation found that either dilating the tricuspid valve opening or displacing the papillary muscles that control its operation can cause the valve to leak. A combination of the two actions can increase the severity of the leakage, which is called tricuspid regurgitation.
Standard clinical procedures that detail when and how tricuspid valve repairs should be performed need to be developed and this study suggests several items that should be considered in developing those protocols, according to the researchers.
In another study published in the journal Circulation: Cardiovascular Imaging, researchers found that the anatomy of the heart’s tricuspid valve can be used to predict the severity of leakage in the valve. Using 3-D echocardiograms from 64 individuals who exhibited assorted grades of tricuspid leakage, the researchers found that pulmonary arterial pressure, the size of the valve opening and papillary muscle position measurements could be used to predict the severity of an individual’s tricuspid regurgitation.
The study will change the focus and direction of future surgical therapies for tricuspid regurgitation to make them better and more durable, the researchers said.
Researchers from the Coulter Department, Emory University, Children’s Hospital Boston and Mount Sinai Medical Center contributed to these two studies.
For more information on this work, visit http://gtresearchnews.gatech.edu/tricuspid-valve-leakage/ and http://gtresearchnews.gatech.edu/tricuspid-regurgitation/.
Research News & Publications Office
Georgia Institute of Technology
75 Fifth Street, N.W., Suite 314
Atlanta, Georgia 30308 USA
Media Relations Contacts: Abby Robinson (abby@innovate.gatech.edu; 404-385-3364) or John Toon (jtoon@gatech.edu; 404-894-6986)
Writer: Abby Robinson
News Contact
Abby Robinson
Research News and Publications
abby@innovate.gatech.edu
404-385-3364
Pagination
- Previous page
- Page 43
- Next page