Sep. 01, 2011
TheParker H. Petit Institute for Bioengineering and Bioscience awarded $100,000 totwo interdisciplinary teams under a new initiative, the PetitBioengineering and Bioscience Collaborative Grant program, which was created to supportearly-stage innovative biotechnology research.
The seed grant recipients address a wide range oftopics including profiling singlecells to understand the heterogeneity of different cell types and newapproaches to traumatic brain injury. Thecall for proposals was welcomed by teams of Petit Institute faculty with onefaculty member from Georgia Tech’s College of Science and one from the Collegeof Engineering.
“This new program aims to promote the collaborationof new teams of researchers and help them establish preliminary results toapply for large external grant proposals,” said Robert Guldberg, PhD, directorof the Petit Institute. “This initiativeis directly in-line with the Petit Institute’s mission, funding cutting-edgeresearch at the interface of bioengineering and the biosciences.”
MelissaKemp, assistant professor in the Wallace H. Coulter Department for BiomedicalEngineering and Greg Gibson, professor in the School of Biology, proposed aproject which aims to develop the measurement tools for relating variability in both genomicand protein information at the single cell level. The ability to conduct this type ofprofiling in single cells represents a remarkable technological advance in thelast two years.
“Studies ofgenomic data often fail to bridge the observed variation in DNA sequences tocellular function, in part due to the variation that is present by both typesof measurement,” Kemp said, “with the technologies this project is developing,we will be able to compare population-averaged data to single cell measurementsin order to gain new insight in relating genes to phenotype.”
MichelleLaPlaca, associate professor in the Wallace H. Coulter Department of BiomedicalEngineering and Al Merrill, professor in the School of Biology, are partneringto merge traumatic brain injury with lipid biology in the hopes of evaluating, for thefirst time, plasma membrane breakdown mechanisms and lipid signaling followingtraumatic brain injury.
“Traumatic brain injury remains a major clinical problem with few effectivetreatments and the devastating sequelae following this type of injury leads tochronic neural deficits,” LaPlaca stated. “We are optimistic that these fundswill propel this important research forward.”
Fundingfor the new seed grants comes chiefly from Petit Institute's endowment as well as contributionsfrom the College of Science and College of Engineering. Each team will receive $50,000 a year for twoyears, however, the second year of funding will be contingent on submission ofan external collaborative grant proposal by July 2012.
News Contact
Marketing Communications Director
Parker H. Petit Institute for Bioengineering and Bioscience
404-385-7001
Aug. 09, 2011
The University System of Georgia Board of Regents today appointed three Georgia Tech faculty members as Regents’ Professors and two as Regents’ Researchers.
The three new Regents’ Professors at Georgia Tech are Mark Prausnitz, professor and director of the Center for Drug Design, Development and Delivery in the School of Chemical & Biomolecular Engineering; Seth Marder, professor in the School of Chemistry and Biochemistry and founding director of the Center for Organic Photonics and Electronics in the colleges of Engineering and Sciences; and Gary Schuster, Vasser Woolley Professor in the School of Chemistry and Biochemistry.
Two Regents’ Researchers appointed include Gisele Bennett, professor and director of the Electro-Optical Systems Laboratory in the Georgia Tech Research Institute; and Suzanne Eskin, principal research scientist in the Wallace H. Coulter Department of Biomedical Engineering at Georgia Tech and Emory University.
“We are immensely proud of our new Georgia Tech Regents’ Professors and Researchers,” said G. P. “Bud” Peterson, Georgia Tech’s president. “They are conducting breakthrough research that is gaining national attention. The fact that we have five Georgia Tech faculty members receiving this honor from the Board of Regents in one year is a reflection of the caliber of scholars we have at Tech.”
A Regents' Professorship and Regents’ Researcher title represents the highest academic status bestowed by the University System of Georgia. It is meant to recognize a substantial, significant and ongoing record of scholarly achievement that has earned high national esteem over a sustained period.
Prausnitz has received international acclaim for his research on biophysical methods of drug delivery, which employ microneedles, ultrasound, lasers, electric fields, heat, convective forces and other physical means to control the transport of drugs, proteins, genes and vaccines into and within the body.
Marder is working on bringing nanotechnology out of the lab and into the marketplace. Using a process known as two-photon absorption, the research groups of Marder and colleague Joseph Perry are developing a broad set of materials for 3D micro- and nanolithography.
Schuster is a nationally known scholar and researcher with an extensive list of published articles on topics ranging from biochemistry through physical chemistry, as well as a number of scientific discoveries with commercial applications. He also held top leadership roles at Georgia Tech such as interim president, provost and dean of the College of Sciences.
Bennett has been praised for the programs she has built around automatic identification technologies using radio frequency identification and container security. Her research activities include the study of optical coherence imaging systems.
Eskin has contributed to research on vascular biology, cardiovascular tissue engineering and gene expression of vascular cells. She studies the comparative effects of mechanical forces accompanying blood flow and pressure on the blood vessel wall.
The titles are awarded by the Board of Regents, which governs the University System of Georgia, upon the unanimous recommendation of the president, the chief academic officer, the appropriate academic dean and three other faculty members named by the president, and upon the approval of the chancellor and the committee on academic affairs.
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
Jun. 16, 2011
AlumnusParker H. “Pete” Petit (ME 1962, MS EM 1964) recently made a commitment toestablish the first endowed director’s chair for a research institute atGeorgia Tech.
TheParker H. Petit Director’s Chair in Bioengineering and Bioscience will be withinthe Parker H. Petit Institute for Bioengineering and Bioscience (IBB), aninterdisciplinary research facility that Petit helped create with an endowment 15years ago.
Thenew endowed director’s chair position is designed to significantly enhanceIBB’s ability to attract and retain an eminent researcher-scholar to thisposition of academic leadership without regard to the academic discipline of the incumbent.
RobertE. Guldberg, director of IBB, will be the first to hold the endowed position. Aprofessor of mechanical engineering, Guldberg was named IBB director in 2009after serving as associate director since 2004. Guldberg is widely recognizedfor his research in biomechanics, biomaterials and tissue engineering with anemphasis on orthopedic applications.
“Pete’spast generosity and good will toward Georgia Tech and IBB catalyzed theformation and growth of a unique and thriving bio-community,” Guldberg said.“Now, he is once again paving the way in support of innovation andinterdisciplinary research by providing this unique endowment—the first of itskind on the Georgia Tech campus.”
Petitis chairman and CEO of Atlanta-based MiMedx Group, an integrateddeveloper, manufacturer and marketer of patent-protected biomaterial-basedproducts focused on augmenting the repair of traumatized tissues andstructures. An exceedingly active alumnus for nearly five decades, Petit serveson the IBB Advisory Board, the Campaign Georgia Tech Steering Committee and theAlexander-Tharpe Fund Board of Trustees.
Petitis also among a handful of Georgia Tech pioneers who recognized very early thevast potential of bioengineering and bioscience, and provided the significantsupport required for that potential to be realized.
Twenty-fiveyears ago, Petit established the Parker H. Petit Distinguished Chair forEngineering in Medicine in the George W. Woodruff School of MechanicalEngineering. In 1995, Petit committed an additional $5 million to establish anendowment for IBB. Petit is once again leading the way in providing vitalsupport for the life sciences at his alma mater.
“What started in the mid 1980s as an initialinvestment on my part has resulted in this amazing program at Georgia Tech,”Petit said. “To see this facility, and the students and faculty and thegroundbreaking interdisciplinary research they’re doing in the facility—there’sno other word for it—it’s overwhelming.”
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
May. 13, 2011
On Saturday, May 21, 2011, the Parker H. PetitInstitute for Bioengineering & Bioscience (IBB) will host itsthird annual Petit Undergraduate Research Scholars fundraising dinner onthe campus of the Georgia Institute of Technology. This event welcomes membersof Atlanta’s business community, university leaders and government officials tocome and support the innovative undergraduate research at IBB.
This year’s dinner will feature guest speakerMitchell H. Gold, MD, who currently serves as president and chief executiveofficer for Dendreon Corporation. Gold will be describing an innovativenew cancer therapy in his presentation entitled, “Winning the War."
Dendreon, a Seattle-based company, recentlycompleted a $70 million Immunotherapy Manufacturing Facility in Union Citywhich brought over 450 jobs to Georgia. Dendreon’s product, Provenge, is thefirst ever FDA-approved immunotherapy for prostate cancer.
The Petit Scholars program began in 2000 as part of a ten-year NationalSciences Foundation grant and has been continued and expanded with the help ofprivate donations. With this year’s fundraising goal of $100,000,sponsorship opportunities are available at platinum, gold, silver and bronzelevels as well as individual dinner ticket purchases.
To date, the Petit Scholars program has provided funding and programsfor 166 students from Georgia Institute of Technology, Emory University,Georgia State University, Morehouse College, Spelman College, Agnes ScottCollege, Gwinnett Technical College and the University of Georgia over the past12 years. IBB Undergraduate Research Scholars program is a competitivescholarship program that allows undergraduates to develop independent researchprojects in Petit Institute laboratories under the director mentorship of agraduate student and faculty member. The programs’ alumni go on to usetheir valuable research experiences in careers as research scientists,academics and physicians.
News Contact
Megan Graziano McDevitt
Marketing and Event Manager
Parker H. Petit Institute for Bioengineering and Bioscience (IBB)
May. 06, 2011
Following a national search,Gary S. May, alumnus, professor and current chair of Electrical and ComputerEngineering, has been appointed as the next dean of Georgia Tech’s College ofEngineering, effective July 1.
“Gary exemplifies the type ofleadership qualities we hope to instill in each of our students,” ProvostRafael L. Bras said. “As a faculty member, administrator and representative ofGeorgia Tech, his impact on his profession and on this institution has beenprofound.”
May will succeed Don Giddens, who will be stepping down as dean of the College of Engineering, a post he has held since 2002, and retiring from the Institute at the end of June.
Asdean, May will assume responsibility for directing the nation’s largestengineering program, one that enrolls nearly 60 percent of Georgia Tech’s studentbody and is home to about half of its tenured and tenure-track faculty.
“Iam grateful for the opportunity to lead a premier institution like the Collegeof Engineering,” said May. “It is truly an honor and a privilege to beentrusted with one of the world’s most respected brands, and I am lookingforward to working with faculty across the college to advance the quality of oureducation and research programs.”
A native of St. Louis,Missouri, May earned his bachelor’s in electrical engineering at Georgia Tech as a student in Georgia Tech's Cooperative Education Program, a five-year accredited, academic program in which students alternate semesters of full-time study with semesters of full-time, paid employment directly related to their major. Current College of Engineering Dean Giddens was also a co-op student at Georgia Tech.
For his graduate studies, May pursued both his master’s and doctoral degrees from the University ofCalifornia, Berkeley. He returned to Tech as an assistant professor in 1991,achieving full professor status in 2000. Two years later he was tapped bythen-President Wayne Clough to serve as his faculty executive assistant, a rolethat introduced him to administrative responsibilities at an institutionallevel.
May,who has chaired the School of Electrical and Computer Engineering since 2005, sharedhis aspirations for the future of the college during a public presentation lastmonth.
“My vision is to create anenvironment where anyone with the aptitude and inclination to study engineeringwill want to come to Georgia Tech,” he said. In partnership with colleagues inthe other colleges, he added, “we will build a community of scholars to addressthe issues and challenges of the world through technology.”
“Gary’srecord of scholarship, his collaborative nature and his tireless mentorship tostudents are admirable,” President Bud Peterson said. “We are very excited about the future of engineeringeducation and research at Georgia Tech under Gary’s leadership.”
Bras thanked the members ofthe search committee for their service, as well as the members of the largercampus community who participated in the evaluation process.
“Weconducted an international search to identify the best possible candidates tolead our largest academic unit,” Bras said. “That the final choice for thismost important and desirable position is one of our own , as a graduate,professor and academic leader, speaks to the excellence of Georgia Tech."
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
Apr. 14, 2011
Don’t miss out on a chance to participate in a unique event thatbrings together public art and science in an innovative way.
“Group Intelligence,” a flash-mob inspired MP3 experience iscoming to Atlanta this month, sponsored by the Out of Hand Theater and the Center for Chemical Evolution.The center is a collaboration of the Georgia Institute of Technology and EmoryUniversity, funded by the National Science Foundation and NASA.
The first “Group Intelligence” flash mob will be held at the Emoryquad at 7 p.m. April 14-16. GroupIntelligence will spread to downtown Atlanta’s Woodruff Park at 7 p.m. April 21-23. Fromthere, the mob will go international to the Oreol Festival in the Netherlandsthis June.
This experiment in the convergence of science and art will helpparticipants understand the behavior of molecules.
Once participants have registered for one of sixperformances, they receive an MP3 track to download, or they sign up to borrowan MP3 player at the event. Following cues on the MP3 track, participantstransform into performers and a spontaneous spectacle unfolds for onlookers.
During the experience, participants travel together, solveproblems, do a little work, have a lot of fun, build something extraordinarytogether and ultimately, achieve "Group Intelligence." How hard they work is upto each individual, but the diversity of the group is key.
“The behavior of a group of molecules can be much morecomplex than the individual molecules alone, much more than the sum of itsparts,” said Martha Grover, Georgia Tech associate professor of chemical and biomolecularengineering and member of the Center for Chemical Evolution. “This isespecially true when the group contains a diverse set of molecules.”
“A similar emergence of complexity occurs in groups ofpeople. In ‘Group Intelligence’, the participants will activelyexperience this important scientific concept. We think this will be moreeffective than simply lecturing about it. We also think it will be morefun,” Grover added.
The “Group Intelligence” performances are free and open tothe public, including children ages 12 and older. Visit the Out of Hand Theaterwebsite to register.
The Center for Chemical Evolution strives to educate thepublic on current scientific theories pertaining to the chemical origins oflife and early evolution.
By participating in “Group Intelligence,” two things will beclear: Order comes out of disorder and chemistryis much more fascinating than we ever imagined.
“All too often, science is seen as dull and irrelevant toour daily lives,” said Meisa Salaita, coordinator for education, outreach anddiversity at the Center for Chemical Evolution. “By working with a grouplike Out of Hand Theater, we are able to work on reversing this bad reputation,bringing our science out to the public in a way that is fun and non-threatening– teaching them about the scientific advances we are making thanks to their taxdollars. “
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
Apr. 04, 2011
Thanks to videoconferencing equipment and a few large-screen televisions, Jennifer Curtis is reaching out to students beyond Tech’s Midtown campus.
Curtis, an assistant professor in the School of Physics, participates in the Direct to Discovery program, a Georgia Tech Research Institute program that brings research labs into K-12 classrooms with a little help from technology.
The program’s goal is to help students better understand various areas of science and mathematics in a way that fosters ongoing interest in these areas.
“Since my lab is so interdisciplinary, we can tie into the curriculum of a physics, chemistry or biology class,” she said.
According to Kimm Bankston, the Winder-Barrow high school teacher Curtis has worked with, the demos have been quite successful and have stimulated student discussions about science that extend beyond the classroom.
“I think the program is an excellent way to inspire the next generation of engineers and scientists,” Curtis added.
Recently, The Whistle had an opportunity to learn more about Curtis and her approach to teaching. Here’s what we learned:
How did you get to Georgia Tech?
In 2006, both my husband and I were seeking tenure-track academic positions. In the end, it was clear that Tech was the best fit for our combined interests both professionally and personally.
How did you become interested in your area of teaching and research?
When I started out as an undergraduate at Columbia University, I wanted to pursue photography and writing. But I experienced a major creative block, which led me back to my first love, science and mathematics. The next semester, I started taking physics classes and the rest is history. As for becoming a biophysicist, I always loved biology and after observing that some of the most interesting work done by physicists was in the area of biophysics, I knew where I needed to be.
In a few sentences, tell us a little bit about your research focus.
My research group studies the mechanics of cells and biomaterials. Also, we invent or develop unique tools to help answer questions about, for example, the coating of a cell.
What is your greatest challenge as an instructor, and how have you dealt with it?
Helping students figure out how to learn and study effectively is always a challenge. For example, there is always a large group of students who work very hard and spend vast amounts of time studying for my introductory physics course. Yet, their performance on tests does not reflect their efforts. I am experimenting with how to instruct students to get to the point where they can internalize and comprehend the difference between deeply understanding how and why they solve problems a certain way versus superficially memorizing or accepting a concept or problem-solving strategy in physics.
What piece of technology could you not live without as an instructor?
I think a tablet PC works wonders for large classroom lecture halls.
Where is the best place to grab lunch and what do you order?
My favorite place used to be Bobby and June’s, but it recently closed. I’d order the Salisbury steak with a side or two of vegetables.
Tell me something unusual about yourself.
When I was younger, I was a competitive épée fencer and trained several hours a day while I was in high school and for part of my time in college.
News Contact
Amelia Pavlik
Communications & Marketing
404-385-4142
Apr. 06, 2011
Two undergraduates from Georgia Tech’s College of Engineering have been named 2011 Goldwater Scholars.
Allison Del Giorno, a sophomore majoring in electrical engineering and minoring in biomedical engineering, and Chun Yong, a junior biomedical engineering student, were awarded Goldwater Scholarships for the 2011-2012 academic year. Del Giorno will also receive the scholarship for her senior year. Each scholarship covers eligible expenses for undergraduate tuition, fees, books, and room and board, up to a maximum of $7,500 annually.
The Barry M. Goldwater Scholarship and Excellence in Education Foundation awarded a total of 275 scholarships to undergraduate sophomores and juniors from the United States. The purpose of the foundation is to provide a continuing source of highly qualified scientists, mathematicians and engineers by awarding scholarships to college students who intend to pursue careers in these fields.
It’s clear why Georgia Tech’s Del Giorno and Yong were recommended for and honored with the Goldwater Scholarship.
Del Giorno, a Georgia Tech President’s Scholar, has held a National Institutes of Health Intramural Research Training Award at the National Institute of Neurological Disorders and Stroke and another at the National Institute of Bioengineering and Biomedical Imaging. She received a Northrop Grumman Engineering Scholarship upon entering Georgia Tech. Del Giorno is studying electrical engineering approaches to the nervous system, specifically investigating the spatiotemporal electrical properties of neurons that control respiration.
Once finished with her undergraduate studies, Del Giorno plans to pursue a doctorate in computational neuroscience to conduct neuroscience research focused on fundamental discoveries for clinical applications.
"I feel so blessed that I was chosen for the Goldwater Scholarship,” said Del Giorno, a native of Eldersburg, Md. “I have amazing family, friends and professors who continue to encourage and guide me as I pursue a career in the neuroscience field."
Another Goldwater Scholar, Yong has received many accolades during his time at Georgia Tech. He was honored as a 2010 Petit Research Scholar and also won several President’s Undergraduate Research Awards. Yong participated in the National Science Foundation’s Research Experiences for Undergraduates and held a Russ Bell Undergraduate Research Scholarship. This summer, he will be attending the Mayo Clinic in Rochester, Minn., for the Summer Undergraduate Research Fellowships program.
Yong is co-founder of a new Biomedical Research and Opportunities Society, executive vice president of the American Medical Student Association and a member of the Biomedical Engineering Society and the National Society of Collegiate Scholars.
In 2003, Yong and his family moved to Suwanee, Ga., after living in Singapore, Vietnam and Malaysia. He is providing for his family as well as doing research, which focuses on insulin-dependent diabetes. He has worked with Professor Athanassios Sambanis’ Lab in the Institute of Bioengineering and Bioscience at Georgia Tech to develop cell-delivering microcapsules for the treatment of diabetes.
In addition to Del Giorno and Yong, Georgia Tech’s Katy Hammersmith, a biomedical engineering sophomore, and Jason Frieman, an aerospace engineering junior, received Honorable Mention from the Barry M. Goldwater Scholarship and Excellence in Education Program.
Georgia Tech can nominate only four students for the Goldwater Scholarship, and it is unusual for all to receive recognition as they did this year, said Karen Adams, interim director of the Fellowship Office at Georgia Tech.
The Barry M. Goldwater Scholarship and Excellence in Education Program was established by Congress in 1986 to honor Sen. Barry M. Goldwater, who served his country for 56 years as a soldier and statesman, including 30 years of service in the U.S. Senate.
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
Mar. 28, 2011
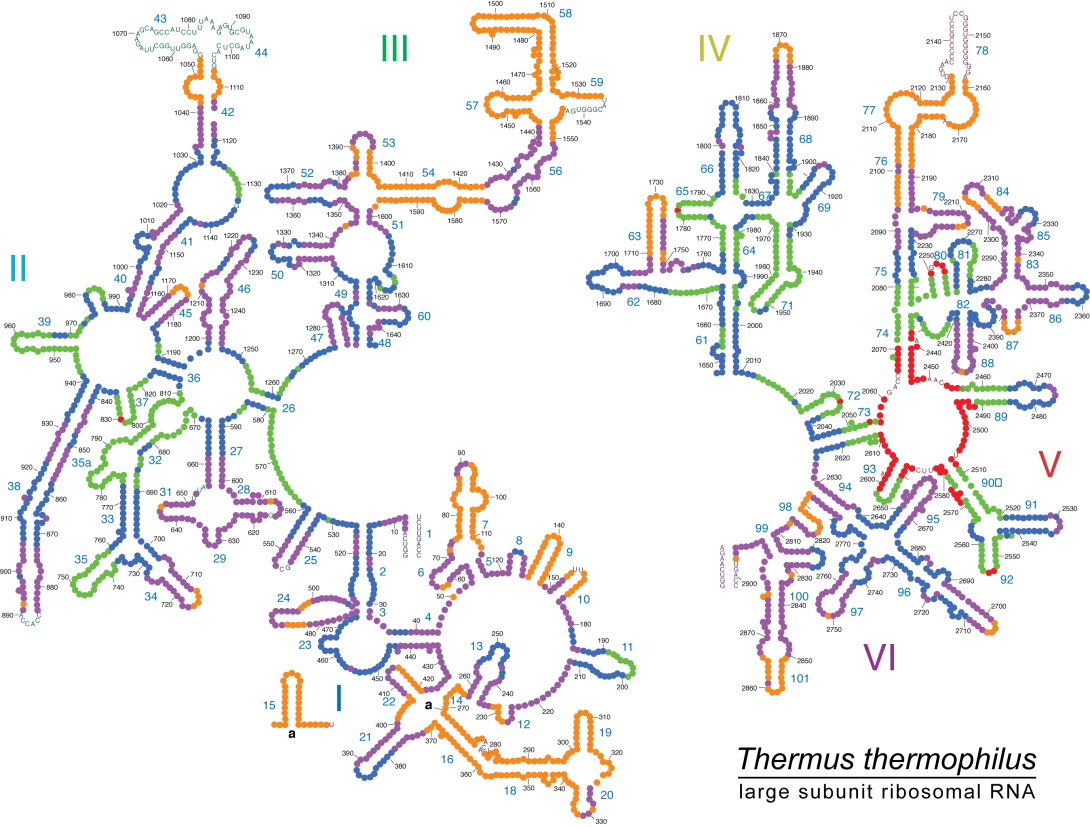
For the 19th year, Georgia Tech is hosting the Suddath Symposium in honor of Leroy "Bud" Suddath, a late professor of the School of Chemistry and Biochemistry. This year's meeting focuses on the ribosome and its structure, function and evolution, exploring scientific implications ranging from understanding the origin and early evolution of life to the development of novel pharmaceuticals. The symposium is organized by the Parker H. Petit Institute for Bioengineering and Bioscience (IBB), the Center for Ribosomal Origins and Evolution, and NASA on April 1-2.
“We are excited that this year's symposium will feature foremost experts on the ribosome, including the 2009 Nobel Laureate in Chemistry – Professor Ada Yonath (Weizmann Institute)," said Adegboyega Oyelere, assistant professor in the School of Chemistry and Biochemistry and the symposium chair.
This annual symposium celebrates the life and contributions of Suddath by discussing the latest developments in the fields of bioengineering and bioscience. The speakers include leading researchers from across the globe. Due to the high quality of the speakers, the 2011 meeting sold out weeks ago.
“If you can not register for the in-person meeting, which is now at capacity, you still have an opportunity to attend virtually. We are using social networking tools for world-wide participation in the meeting,“ said co-organizer, Loren Williams, professor in the School of Chemistry and Biochemistry and director of the Center for Ribosomal Origins and Evolution.
Georgia Tech has partnered with NASA to showcase the symposium, in its entirety, over the internet. Virtual participants will view the presentations in real-time and can chat with the other cyber-attendees using Facebook on the home page of the symposium's website. Over 200 attendees, more than twice the number that will attend in person, from around the globe have registered to participate virtually. Countries represented include Australia, Brazil, Bulgaria, Canada, Chile, Colombia, Croatia, Denmark, Estonia, Germany, India, Iran, Ireland, Japan, Mexico, Serbia, Spain, Sweden, Switzerland, Tanzania, Turkey, Portugal and Venezuela.
“IBB will be showcased around the world as people beam into the seminar room to see our local symposium worldwide,” Williams said.
The ribosome is a molecular machine that is responsible for protein synthesis in all living cells. This indispensable component of life, which contains both RNA and proteins, can be viewed as a molecular fossil. That is, the comparison of ribosomal RNA and proteins from distantly related organisms suggests that the origins and evolution of protein synthesis remain imprinted in present day ribosomes, providing a “rewindable” molecular recording of early evolution that appears to go all the way back to the origin of life. Because the ribosome is central to the biochemistry of all life, it is a major target for drug development.
Each year, the Suddath Symposium theme changes, although the scientific committee selects an interdisciplinary topic that they feel Suddath would have been excited about. "A symposium focusing on the ribosome is particularly fitting, as Bud (Suddath) contributed to solving the structure of tRNA, a key substrate which is used by the ribosome to make proteins,” Oyelere explained. Suddath’s research efforts ultimately led to a set of protein growth experiments aboard the Space Shuttle in 1988.
News Contact
Megan Graziano McDevitt
Marketing and Event Manager
Parker H. Petit Institute for Bioengineering and Bioscience (IBB)
(404) 385-7001
Mar. 10, 2011
As Georgia Tech celebrates the 50th anniversary ofthe matriculation of African-American students, the Institute is proud of theachievements of the FACES program in bringing diversity to science andengineering education.
Since 1998, more than 300 minority students earned theirdoctorate in science, technology, engineering and math, thanks in part to theFACES program.
Georgia Tech ranked no. 1 in the U.S. last year for awardingthe most engineering doctoral degrees to African-American students and allminority students, according to DiverseIssues in Higher Education.
Georgia Tech oversees the FACES program, which stands forFacilitating Academic Careers in Engineering and Science, in partnership withEmory University, Morehouse College and Spelman College. The National ScienceFoundation funds the program.
“Over the last decade, the FACES program has contributedsignificantly to the formation of an environment at Georgia Tech in which thecompletion of a STEM doctorate and consideration of an academic career arevalued by talented minority students and supported by the campus," saidGary May, Georgia Tech’s Steve W. Chaddick School Chair of the School ofElectrical and Computer Engineering. "We are very proud of what has beencreated here and look forward to the contributions of our students as theypursue their careers."
A shining example of the FACES program’s success is ManuPlatt, assistant professor of biomedical engineering in the Wallace H. CoulterDepartment of Biomedical Engineering at Georgia Tech and Emory University.
Platt not only received a FACES grant that allowed him topursue professional development while a postdoctoral fellow at M.I.T., but alsoreceived the FACES Career Initiation Grant when he joined the Georgia Techfaculty in 2009 to kick-start his research.
Platt, who this fall received a $1.5 million NIH Director'sNew Innovator Award to support his research on reducing stroke in children withsickle cell disease, said the prestige of being a FACES recipient has enriched hiscareer.
“Georgia Tech is a special place to be such a top-qualityengineering school and to have a diverse faculty,” Platt said. “I probably wouldn’t be as happy as aprofessor as I am today without the FACES program. Truly, I wouldn’t have theopportunity to be mentored by the African-American professors who helped mebuild a community and have showed me how to avoid the brick walls that youinevitably run into."
Today, Platt sits on the FACES steering committee thatoversees the program and helps mentor African-American undergraduate andgraduate students from Georgia Tech, Emory, Morehouse and Spelman, who willbecome the next generation of STEM professors.
“I enjoy just talking to them and selling them on why beinga professor is so great,” Platt said. “You get to see students develop, learnand grow, and eventually move forward with their career.”
The FACES program includes three components:
- The Summer Undergraduate ResearchEngineering/Science (SURE) program seeks to motivate African-Americans to entergraduate school. Students of at least junior level are recruited on anationwide basis and paired with both a faculty and a graduate mentor toundertake research projects. SURE students are housed on campus and areprovided with a $5,000 stipend, $600 travel allowance and a meal plan.
- For graduate students who are committed topursuing doctorates, the FACES Fellowship provides an add-on stipend of either$3,000 or $5,000 per year, depending on the status of the student's Ph.D.candidacy. In addition to receivingmonetary support, the FACES Fellows participate in workshops designed to helpthem excel in graduate school and prepare them for careers in research andacademia.
- The FACES program also supports future facultydevelopment. Each year, one promisingscientist who accepts a postdoctoral position in engineering or science at anyU.S. college or university is awarded a $35,000 grant, money that he or she cantake with them, as Platt did. FACES also awards two $30,000 Career InitiationGrants to doctoral students who accept a tenure track faculty position in anengineering or science-related field at a U.S. college or university.
All of the pieces of FACES contribute to the program’ssuccess, May said.
“At Georgia Tech, we’ve seen that a key factor for motivatingstudents to pursue advanced degrees and research careers in science andengineering is fruitful research experiences,” he said. “Quality interactionswith engineering faculty can have a significant impact on a student’s decisionto pursue graduate education.”
News Contact
Georgia Tech Media Relations
Laura Diamond
laura.diamond@comm.gatech.edu
404-894-6016
Jason Maderer
maderer@gatech.edu
404-660-2926
Pagination
- Previous page
- Page 13
- Next page