Jun. 12, 2024
Adoptive T-cell therapy has revolutionized medicine. A patient’s T-cells — a type of white blood cell that is part of the body’s immune system — are extracted and modified in a lab and then infused back into the body, to seek and destroy infection, or cancer cells.
Now Georgia Tech bioengineer Ankur Singh and his research team have developed a method to improve this pioneering immunotherapy.
Their solution involves using nanowires to deliver therapeutic miRNA to T-cells. This new modification process retains the cells’ naïve state, which means they’ll be even better disease fighters when they’re infused back into a patient.
“By delivering miRNA in naïve T cells, we have basically prepared an infantry, ready to deploy,” Singh said. “And when these naïve cells are stimulated and activated in the presence of disease, it’s like they’ve been converted into samurais.”
Lean and Mean
Currently in adoptive T-cell therapy, the cells become stimulated and preactivated in the lab when they are modified, losing their naïve state. Singh’s new technique overcomes this limitation. The approach is described in a new study published in the journal Nature Nanotechnology.
“Naïve T-cells are more useful for immunotherapy because they have not yet been preactivated, which means they can be more easily manipulated to adopt desired therapeutic functions,” said Singh, the Carl Ring Family Professor in the Woodruff School of Mechanical Engineering and the Wallace H. Coulter Department of Biomedical Engineering.
The raw recruits of the immune system, naïve T-cells are white blood cells that haven’t been tested in battle yet. But these cellular recruits are robust, impressionable, and adaptable — ready and eager for programming.
“This process creates a well-programmed naïve T-cell ideal for enhancing immune responses against specific targets, such as tumors or pathogens,” said Singh.
The precise programming naïve T-cells receive sets the foundational stage for a more successful disease fighting future, as compared to preactivated cells.
Giving Fighter Cells a Boost
Within the body, naïve T-cells become activated when they receive a danger signal from antigens, which are part of disease-causing pathogens, but they send a signal to T-cells that activate the immune system.
Adoptive T-cell therapy is used against aggressive diseases that overwhelm the body’s defense system. Scientists give the patient’s T-cells a therapeutic boost in the lab, loading them up with additional medicine and chemically preactivating them.
That’s when the cells lose their naïve state. When infused back into the patient, these modified T-cells are an effective infantry against disease — but they are prone to becoming exhausted. They aren’t samurai. Naïve T-cells, though, being the young, programmable recruits that they are, could be.
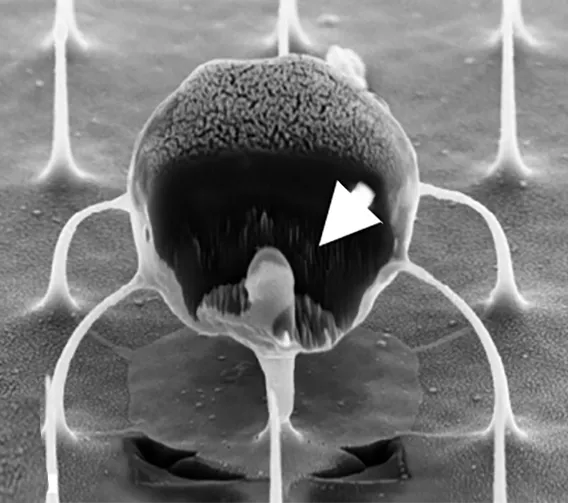
The question for Singh and his team was: How do we give cells that therapeutic boost without preactivating them, thereby losing that pristine, highly suggestable naïve state? Their answer: Nanowires.
NanoPrecision: The Pointed Solution
Singh wanted to enhance naïve T-cells with a dose of miRNA. miRNA is a molecule that, when used as a therapeutic, works as a kind of volume knob for genes, turning their activity up or down to keep infection and cancer in check. The miRNA for this study was developed in part by the study’s co-author, Andrew Grimson of Cornell University.
“If we could find a way to forcibly enter the cells without damaging them, we could achieve our goal to deliver the miRNA into naïve T cells without preactivating them,” Singh explained.
Traditional modification in the lab involves binding immune receptors to T-cells, enabling the uptake of miRNA or any genetic material (which results in loss of the naïve state). “But nanowires do not engage receptors and thus do not activate cells, so they retain their naïve state,” Singh said.
The nanowires, silicon wafers made with specialized tools at Georgia Tech’s Institute for Electronics and Nanotechnology, form a fine needle bed. Cells are placed on the nanowires, which easily penetrate the cells and deliver their miRNA over several hours. Then the cells with miRNA are flushed out from the tops of the nanowires, activated, eventually infused back into the patient. These programmed cells can kill enemies efficiently over an extended time period.
“We believe this approach will be a real gamechanger for adoptive immunotherapies, because we now have the ability to produce T-cells with predictable fates,” says Brian Rudd, a professor of immunology at Cornell University, and co-senior author of the study with Singh.
The researchers tested their work in two separate infectious disease animal models at Cornell for this study, and Singh described the results as “a robust performance in infection control.”
In the next phase of study, the researchers will up the ante, moving from infectious disease to test their cellular super soldiers against cancer and move toward translation to the clinical setting. New funding from the Georgia Clinical & Translational Science Alliance is supporting Singh’s research.
CITATION: Kristel J. Yee Mon, Sungwoong Kim, Zhonghao Dai, Jessica D. West, Hongya Zhu5, Ritika Jain, Andrew Grimson, Brian D. Rudd, Ankur Singh. “Functionalized nanowires for miRNA-mediated therapeutic programming of naïve T cells,” Nature Nanotechnology.
FUNDING: Curci Foundation, NSF (EEC-1648035, ECCS-2025462, ECCS-1542081), NIH (5R01AI132738-06, 1R01CA266052-01, 1R01CA238745-01A1, U01CA280984-01, R01AI110613 and U01AI131348).
News Contact
Jerry Grillo
Jul. 06, 2023
Now testing on the research site locally for over writing.
10:55 am
Needing to update this article to see the auto pull and notice ios the Infinite check box gets overwritten.
september 2, 2025
Bacon ipsum dolor amet pork loin swine cupim turkey landjaeger hamburger turducken picanha porchetta chislic t-bone venison shank. Jerky chislic beef strip steak, flank corned beef pork loin short loin sausage. Spare ribs venison kevin, ham hock swine pastrami hamburger shoulder sirloin ground round cupim tail brisket salami jowl. T-bone turkey meatball chislic cupim, shoulder ground round ball tip meatloaf flank alcatra pastrami leberkas pork loin. Landjaeger capicola shank turducken kielbasa, turkey rump hamburger tri-tip. Turkey short loin leberkas picanha ham hock.
Cupim pork loin flank shoulder burgdoggen. Hamburger burgdoggen t-bone tongue cupim. Pork loin frankfurter boudin tail, pancetta meatloaf pork chicken filet mignon cupim brisket alcatra. Fatback t-bone alcatra, cow filet mignon brisket hamburger picanha spare ribs. Tenderloin turducken sausage shoulder, brisket ground round tri-tip tongue chuck pork chop ball tip t-bone buffalo prosciutto. Boudin ball tip buffalo, pork loin bresaola leberkas pancetta salami doner. Pork chop tri-tip andouille beef tongue filet mignon strip steak.
Corned beef t-bone ball tip, sausage ham hock hamburger chuck strip steak shankle beef frankfurter chicken pork belly tenderloin. Frankfurter tenderloin ribeye pork chop, t-bone pork belly beef jerky shoulder. Cupim short ribs jerky drumstick. Tongue venison turducken landjaeger meatball pork loin burgdoggen ribeye spare ribs. Salami pork loin chicken capicola.
News Contact
Tim Whelan
tim.whelan@research.gatech.edu
Web Dev Lead
Jul. 20, 2021
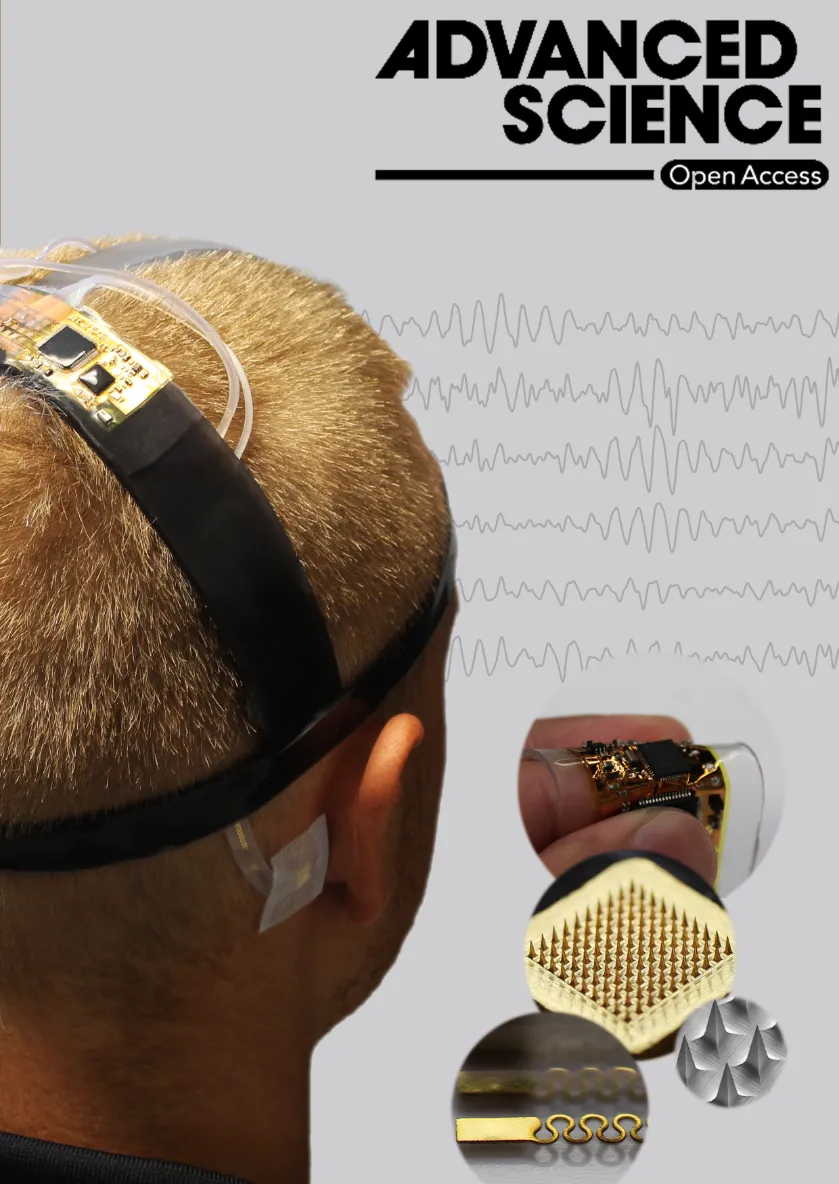
A new wearable brain-machine interface (BMI) system could improve the quality of life for people with motor dysfunction or paralysis, even those struggling with locked-in syndrome – when a person is fully conscious but unable to move or communicate.
A multi-institutional, international team of researchers led by the lab of Woon-Hong Yeo at the Georgia Institute of Technology combined wireless soft scalp electronics and virtual reality in a BMI system that allows the user to imagine an action and wirelessly control a wheelchair or robotic arm.
The team, which included researchers from the University of Kent (United Kingdom) and Yonsei University (Republic of Korea), describes the new motor imagery-based BMI system this month in the journal Advanced Science.
“The major advantage of this system to the user, compared to what currently exists, is that it is soft and comfortable to wear, and doesn’t have any wires,” said Yeo, associate professor on the George W. Woodruff School of Mechanical Engineering.
BMI systems are a rehabilitation technology that analyzes a person’s brain signals and translates that neural activity into commands, turning intentions into actions. The most common non-invasive method for acquiring those signals is ElectroEncephaloGraphy, EEG, which typically requires a cumbersome electrode skull cap and a tangled web of wires.
These devices generally rely heavily on gels and pastes to help maintain skin contact, require extensive set-up times, are generally inconvenient and uncomfortable to use. The devices also often suffer from poor signal acquisition due to material degradation or motion artifacts – the ancillary “noise” which may be caused by something like teeth grinding or eye blinking. This noise shows up in brain-data and must be filtered out.
The portable EEG system Yeo designed, integrating imperceptible microneedle electrodes with soft wireless circuits, offers improved signal acquisition. Accurately measuring those brain signals is critical to determining what actions a user wants to perform, so the team integrated a powerful machine learning algorithm and virtual reality component to address that challenge.
The new system was tested with four human subjects, but hasn’t been studied with disabled individuals yet.
“This is just a first demonstration, but we’re thrilled with what we have seen,” noted Yeo, Director of Georgia Tech’s Center for Human-Centric Interfaces and Engineering under the Institute for Electronics and Nanotechnology, and a member of the Petit Institute for Bioengineering and Bioscience.
New Paradigm
Yeo’s team originally introduced soft, wearable EEG brain-machine interface in a 2019 study published in the Nature Machine Intelligence. The lead author of that work, Musa Mahmood, was also the lead author of the team’s new research paper.
“This new brain-machine interface uses an entirely different paradigm, involving imagined motor actions, such as grasping with either hand, which frees the subject from having to look at too much stimuli,” said Mahmood, a Ph. D. student in Yeo’s lab.
In the 2021 study, users demonstrated accurate control of virtual reality exercises using their thoughts – their motor imagery. The visual cues enhance the process for both the user and the researchers gathering information.
“The virtual prompts have proven to be very helpful,” Yeo said. “They speed up and improve user engagement and accuracy. And we were able to record continuous, high-quality motor imagery activity.”
According to Mahmood, future work on the system will focus on optimizing electrode placement and more advanced integration of stimulus-based EEG, using what they’ve learned from the last two studies.
This research was supported by the National Institutes of Health (NIH R21AG064309), the Center Grant (Human-Centric Interfaces and Engineering) at Georgia Tech, the National Research Foundation of Korea (NRF-2018M3A7B4071109 and NRF-2019R1A2C2086085) and Yonsei-KIST Convergence Research Program. Georgia Tech has a pending patent application related to the work described in this paper.
Citation: Musa Mahmood, et al., “Wireless Soft Scalp Electronics and Virtual Reality System for Motor Imagery-based Brain-Machine Interfaces.” (Advanced Science, July 2021)
Links
Center for Human-Centric Interfaces and Engineering
Petit Institute for Bioengineering and Bioscience
George W. Woodruff School of Mechanical Engineering
Aug. 06, 2020
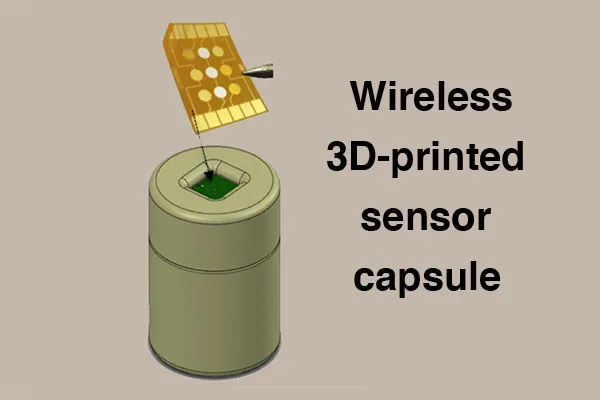
BioFabUSA, a Department of Defense-funded Manufacturing Innovation Institute within the Manufacturing USA network, has awarded the Georgia Institute of Technology and industry partner Rockwell Automation a project entitled, “Wireless Electrochemical Sensor Capsules for Real-Time Monitoring of Cell Secretomes and Culture Media in Tissue Growth Bioreactors.” Real-time bioprocess monitoring and control is needed for the scalable production and deployment of efficacious tissue engineered medical products (TEMPs) at reasonable cost.
Billyde Brown, the project's principal investigator, explained, “we are addressing this challenge by working with BioFabUSA, our partners at the Georgia Tech School of Materials Science and Engineering, the Marcus Center for Therapeutic Cell Characterization and Manufacturing, as well as Rockwell Automation, to develop a fully integrated, wireless, 3D-printed sensor ‘capsule’ to be used for in-situ multiplexed monitoring of critical quality attributes (CQAs). The targeted CQAs include pH, glucose, lactate, and select secreted biomarker concentrations from human mesenchymal stem cells – one of the most common cell types used in tissue engineering.”
In both biopharmaceutical and regenerative medicine industries, an urgent need remains for in-line sensor technology for quantitative real-time bioprocess monitoring and control. Unfortunately, many key CQAs are still monitored off-line or at-line using destructive testing or technologies of significant complexity and cost. In at-line measurement, the sample is typically withdrawn from a single location in the bioreactor and analyzed in close proximity to the process stream, whereas in off-line measurements, the sample is taken to a laboratory and the results are usually not returned in a timely manner for process control.
The Georgia Tech team has previously developed potentiometric sensors based on an extended gate field-effect-transistor (FET) topology whereby a separate gold electrode surface is functionalized with an analyte-specific layer that selectively reacts or binds with the chemical or biomolecule of interest. The charge associated with the attached analyte results in a potential change of the gold electrode. These sensors have previously been used to detect chemicals such as pH and lactate, as well as specific proteins/antibodies in a laboratory environment with accuracy and dynamic range equivalent to Surface Plasmon Resonance (SPR) and Enzyme-Linked Immunosorbent Assay (ELISA). One of the unique aspects of this system is that each sensor surface can be individually functionalized permitting multiplexed (simultaneous) detection of almost any number of different chemicals/biomolecules of interest.
In this project, the Georgia Tech team will integrate these sensors into a “capsule” device smaller than the size of a golf-ball and packaged in a 3D-printed waterproof and biocompatible polymer. The capsule will contain a multiplexed sensor chip, with sealed opening to facilitate interaction between the sensor chip and tissue culture environment, Li-polymer battery, and electronics for micro-control, data acquisition and wireless transmission of sensor data to the smartphone of a technician in charge of monitoring the bioreactor process. In addition, Georgia Tech will work with Rockwell to develop an IoT platform such that other permitted internet-connected devices can securely access the data via a cloud server. Another unique aspect of this technology is that multiple “capsules” could be deployed within a stirred tank bioreactor during high volume production of medical products with the ability to move efficiently throughout the bioreactor due to the mechanical forces of the impellors. This would allow for unprecedented simultaneous measurements at various points within the bioreactor, giving accurate representations of the homogeneity of key parameters over time thus achieving in-situ monitoring of CQAs with high spatial and temporal resolution.
Georgia Tech project leads include Billyde Brown, Ph.D., Kan Wang, Ph.D., and Eric Vogel, Ph.D. Brown is research faculty and director of manufacturing education programs at the Georgia Tech Manufacturing Institute (GTMI). Wang is lead researcher of additive manufacturing in the Bio-Engineering Research Laboratory at GTMI. Vogel is a professor at the School of Materials Science and Engineering and deputy director for the Institute of Electronics and Nanotechnology at Georgia Tech. The Georgia Tech project leads will also receive support and assistance from Carolyn Yeago, Ph.D., and Krishnendu Roy, Ph.D. whom are directors of the Marcus Center for Therapeutic Cell Characterization and Manufacturing (MC3M). Leading the project for Rockwell Automation is Wayne Charest, who also serves as a liaison between Rockwell and BioFabUSA.
“Being able to obtain real-time data on relevant biomarkers will be critical in advancing the field of tissue engineering,” said Stephanie Robichaud, technical project manager with the Advanced Regenerative Manufacturing Institute. “Getting this important information and being able to react to it quickly will result in more consistent manufacturing of a final product that meets its critical quality attributes.”
About the Georgia Institute of Technology
The Georgia Institute of Technology, also known as Georgia Tech, is one of the nation’s leading research universities — a university that embraces change while continually Creating the Next. The next generation of leaders. The next breakthrough startup company. The next lifesaving medical treatment.
Georgia Tech provides a focused, technologically based education to more than 36,000 undergraduate and graduate students. The Institute has many nationally recognized programs, all top-ranked by peers and publications alike, and is ranked among the nation’s top five public universities by U.S. News & World Report. It offers degrees through the Colleges of Computing, Design, Engineering, Sciences, the Scheller College of Business, and the Ivan Allen College of Liberal Arts. As a leading technological university, Georgia Tech has more than 100 centers focused on interdisciplinary research that consistently contribute vital research and innovation to American government, industry, and business. https://www.gatech.edu/
About Rockwell Automation
Rockwell Automation is the largest company in the world that is dedicated to industrial automation and information and is committed to enabling the next generation of smart manufacturing. Rockwell’s mission is to improve the quality of life by making the world more productive and sustainable.
https://www.rockwellautomation.com
About BioFabUSA
BioFabUSA is a DOD-funded Manufacturing USA Innovation Institute (MII) sustained by the Advanced Regenerative Manufacturing Institute (ARMI), a non-profit organization located in Manchester, New Hampshire. ARMI's mission is make practical the scalable, consistent, cost-effective manufacturing of tissue engineered medical products and tissue-related technologies, to benefit existing industries and grow new ones. https://www.armiusa.org/
Georgia Tech Manufacturing Institute
813 Ferst Drive, NW
Atlanta, GA 30332 USA
Media Relations Contact: Walter Rich (walter.rich@research.gatech.edu)
News Contact
Aug. 06, 2020
BioFabUSA, a Department of Defense-funded Manufacturing Innovation Institute within the Manufacturing USA network, has awarded the Georgia Institute of Technology and industry partner, Akron Biotech, a project titled, “Supply Chain and Process Modeling Algorithms, Methods, and Tools for Tissue Manufacturing and Distribution”. This project will address significant national supply chain issues related to distributing tissue engineered medical products (TEMPs) to U.S. patients in need.
The project aims to create the first simulation-based supply chain model for the rapidly evolving and future facing TEMPs industry, to minimize manufacturing and logistics costs and risks, incorporate Department of Defense (DOD) and other stakeholders’ perspectives into supply chain modeling, inform standards development, and support workforce development.
“Having a supply chain model will be instrumental in helping new and existing companies plan for the most efficient process flows, resource usage, and cost savings,” said Stephanie Robichaud, technical project manager with the Advanced Regenerative Manufacturing Institute. “Many startup companies do not realize some of the intricacies in managing their supply chain and many established companies realize the importance of it after experiencing inefficiencies. Having a model that these companies can use will help advance the field of tissue engineering as they plan for scale-up.”
According to Ben Wang, executive director of the Georgia Tech Manufacturing Institute (GTMI) and professor in the Stewart School of Industrial and Systems Engineering, “hundreds if not thousands of patients are waiting for tissues and organs in order to have a normal healthy life. Our project is a bold initiative to democratize distribution of replacement tissues and organs by streamlining national supply chains. This project will develop simulation-based tools to enhance the efficiency and resilience of the TEMPs supply chain, making these personalized medicines more affordable and more accessible.”
The growth of the TEMP industry is going to change the supply chain of medical tissues disruptively. To embrace this change, a system-level decision support tool is essential for adopting more cost-effective manufacturing processes and making better investment decisions. To ensure successful commercialization and adoption of this new supply chain decision support tool, the project team will engage multiple stakeholders including DOD, government, regulatory bodies, standards setting organizations, patients, industry, academia, policy experts, education and workforce development experts.
Georgia Tech project leads include Ben Wang, Ph.D., Chelsea C. White III, Ph.D, and Kan Wang, Ph.D. Ben Wang is Gwaltney Chair in Manufacturing Systems, professor in the Stewart School of Industrial & Systems Engineering and School of Materials Science and Engineering at Georgia Tech. In addition, he serves as executive director of the Georgia Tech Manufacturing Institute (GTMI). Chelsea C. White III is the Schneider National Chair in Transportation and Logistics and professor in the H. Milton Stewart School of Industrial and Systems Engineering at Georgia Tech. Kan Wang is lead researcher of additive manufacturing in the Bio-Engineering Research Laboratory at GTMI.
Leading the project for Akron Biotech is Ezequiel Zylberberg, Ph.D, who is vice president of product development and planning. According to Ezequiel, “the future of regenerative medicine depends on more than our ability to address the scientific challenges of generating the next generation of advanced therapies. Advancing these novel treatments in a way that is scalable will require significant advances in manufacturing innovation. We are eager to collaborate with our colleagues at Georgia Tech, at BioFab USA, and throughout the regenerative medicine industry to confront the challenge of scalability and supply chain resilience through this modelling effort.”
About the Georgia Institute of Technology
The Georgia Institute of Technology, also known as Georgia Tech, is one of the nation’s leading research universities — a university that embraces change while continually Creating the Next. The next generation of leaders. The next breakthrough startup company. The next lifesaving medical treatment.
Georgia Tech provides a focused, technologically based education to more than 36,000 undergraduate and graduate students. The Institute has many nationally recognized programs, all top-ranked by peers and publications alike, and is ranked among the nation’s top five public universities by U.S. News & World Report. It offers degrees through the Colleges of Computing, Design, Engineering, Sciences, the Scheller College of Business, and the Ivan Allen College of Liberal Arts. As a leading technological university, Georgia Tech has more than 100 centers focused on interdisciplinary research that consistently contribute vital research and innovation to American government, industry, and business. https://www.gatech.edu/
About Akron Biotech
Akron is a leading materials manufacturer and services provider to the regenerative medicine industry, accelerating the development and commercialization of advanced therapies. Founded in 2006, Akron is an ISO 13485-certified company that operates in line with cGMPs and international standards, enabling advanced therapy developers to de-risk their supply chains and facilitate regulatory approval. The company's unique business model emphasizes knowledge, flexibility and unparalleled service—from development through to commercialization. For more information, please visit www.akronbiotech.com.
About BioFabUSA
BioFabUSA, is a DOD-funded Manufacturing USA Innovation Institute (MII) sustained by the Advanced Regenerative Manufacturing Institute (ARMI) is a non-profit organization located in Manchester, New Hampshire. ARMI's mission is to make practical the scalable, consistent, cost-effective manufacturing of tissue engineered medical products and tissue-related technologies, to benefit existing industries and grow new ones. https://www.armiusa.org/
Georgia Tech Manufacturing Institute
813 Ferst Drive, NW
Atlanta, GA 30332 USA
Media Relations Contact: Walter Rich (walter.rich@research.gatech.edu)
News Contact
May. 01, 2012
Grad students! Minoring in policy? Want to take something really different, but relevant to your technical training, this summer? Want to take a SUMMER course?
In the SUMMER 2012 session Margaret Kosal, PhD, (INTA professor) and Robert Butera, PhD, are offering this course in Biotechnology and International Affairs which is cross-listed between BMED and INTA.
NOTE: This is a 5 week session, taught 3 hours/day 3 days/week for 5 weeks. Take note of the dates/days when registering!
INTA 8803 MK / BMED 8813 BIS
Biotechnology and International Security
This course will explore the interface between biotechnology and national security concerns. Rapid biotechnological changes are anticipated to occur over the ensuing decades in a globalized world characterized by complex security challenges. What security concerns are posed by rapid developments in biotechnology? How do governments deal with these concerns? Can regulatory frameworks keep pace with rapid developments in biotechnology? How are these issues handled at an international level? We will consider the role of government and non-governmental organizations in efforts to control these technologies. Finally, we will examine the role of the industry and the open market in shaping policy on these technologies.
News Contact
Apr. 13, 2012
Applications are being sought for the 2012-2013 Robert M. Nerem International Travel Award. This award was endowed in 2005 by friends and colleagues of Nerem's to honor his life-long contributions in the bioengineering and bioscience field and encourage predoctoral and postdoctoral trainees to broaden their research experiences by establishing an international collaboration and traveling to an international destination.
The award provides up to $3,000 for the selected applicant to travel abroad with preference given to those who will learn new tools or techniques. To be eligible for the award the trainee must have one year remaining in their research and complete their travel by August 31, 2013. For the 2012-2013 award, the applications are due May 11, 2012.
As the Petit Institute’s founding director, Bob passionately served the community for 14 years and successfully led the institute to national and international prominence in the fields of bioengineering & bioscience.
Everyone that knows Bob, knows he loves to travel. His travels have brought him to all corners of the world and it is through his travel that he has served as a great champion of Georgia Tech and the biocommunity as a whole.
The Nerem International Travel Award has allowed trainees an opportunity to travel to a wide variety of international universities and research institutes, including the Karolinska Institute, Stockholm, Sweden; RIKEN Brain Science Institute, Japan; the National University of Singapore; University of Twente, The Netherlands; Queensland University of Technology, Australia; and Consorzio Interuniversitario Lombardo per L’Elaborazione Automatica, Milan, Italy.
Nerem came to Georgia Tech in the winter of 1987 as a professor in the School of Mechanical Engineering and as the Parker H. Petit Distinguished Chair for Engineering in Medicine. He is one of the grandfathers of the booming bio-community that exists on campus today. Prior to coming to Georgia Tech, he was a professor and chairman in the Department of Mechanical Engineering at the University of Houston from 1979 to 1986 and on the faculty at the Ohio State University form 1964 to 1979.
Apr. 13, 2012
April 13, 2012 – Georgia Institute of Technology is hosting the fifth biennial Astrobiology Science Conference (AbSciCon), April 16-20 at the Georgia Tech Hotel and Conference Center in Atlanta. Loren Williams, Ph.D., professor, School of Chemistry and Biochemistry and Eric Gaucher, Ph.D., associate professor, School of Biology at Georgia Tech are the co-chairs of the conference.
AbSciCon attracts a community of scientists working in the multidisciplinary field of astrobiology – the study of the origin, evolution, distribution, and future of life in the universe – and highlights research supported by NASA's Astrobiology Program.
NASA’s Astrobiology program addresses three fundamental questions: How does life begin and evolve? Is there life beyond Earth and, if so, how can we detect it? What is the future of life on Earth and in the universe?
In striving to answer these questions and improve understanding of biological, planetary, cosmic phenomena and relationships among them, experts will discuss astrobiology research to help advance laboratory and field research into the origins and early evolution of life on Earth and studies of the potential for life to adapt to challenges on Earth and in space.
A record number of abstracts (more than 800) were accepted for this meeting, and the scientific program is packed with talks on current research. Among hot topics on the AbSciCon 2012 agenda are Mars exploration and the Mars Science Laboratory mission, current research on extrasolar planet habitability and latest results from analyses of extraterrestrial materials such as meteorites and comet dust samples. All plenary sessions and four selected technical sessions will be webcast live.
One highlight of the conference will be the final round of the NASA Astrobiology Program’s first annual Famelab Astrobiology science communication competition, April 16 at 7 pm. Nichelle Nichols, known for her portrayal of Lt. Uhura in the original “Star Trek” television series, will be hosting this public event which also will be webcast live. Other highlights include a welcome reception at the Parker H. Petit Institute for Bioengineering and Bioscience at Georgia Tech on Sunday April 15, 5-7 p.m., and conference dinner at the Georgia Aquarium on Wednesday April 18, 5-9 p.m.
News Contact
Megan Graziano McDevitt
Marketing Communications Director
Parker H. Petit Institute for Bioengineering & Bioscience
404-385-7001
Linda Billings
School of Media and Public Affairs
George Washington University
Chair, AbSciCon 2012 Communications Committee
Apr. 11, 2012
April 10, 2012 – Georgia Institute of Technology will host the Famelab Astrobiology Finals, Monday April 16, 7-9 p.m., in the Grand Ballroom of the Georgia Tech Hotel and Conference Center. The event is free and open to the public.
Famelab Astrobiology is a science communication competition intended to encourage up-and-coming new scientists to hone their skills in communicating complex scientific concepts to public audiences.
Nichelle Nichols, known for her portrayal of Lt. Uhura in the original “Star Trek” television series, will host this event, which also will be webcast live and broadcast on NASA TV.
FameLab Astrobiology finalists will have three minutes to explain a science topic of their choice to a public audience – no slides, no charts, and only props they can carry onstage. A panel of experts in science and science communication will judge the competition.
Since January more than 70 early-career astrobiologists have competed in Famelab Astrobiology preliminary competitions in Houston, Denver, Washington, D.C., and online. The 10 finalists, from all over the country, will compete in the Atlanta finals. The winner in Atlanta will compete in International FameLab’s final competition in the U.K. this summer.
The Atlanta finals are the culmination of the first annual Famelab Astrobiology competition, sponsored by NASA’s Astrobiology Program. Famelab Astrobiology, an offshoot of International FameLab, aims to provide experience and training in science communication to the next generation of astrobiologists.
The FameLab Astrobiology competition is being held in conjunction with AbSciCon 2012, an astrobiology science conference with over 750 attendees taking place on the campus of Georgia Tech. Loren Williams, Ph.D., professor in the School of Chemistry and Biochemistry and Eric Gaucher, Ph.D., associate professor from the School of Biology at Georgia Tech are the co-chairs of the conference. The AbSciCon conference, held every two years, focuses on the multidisciplinary field of astrobiology – the study of the origin, evolution, distribution, and future of life in the universe – and highlights research supported by NASA's Astrobiology Program.
AbSciCon 2012 on Twitter: https://twitter.com/#!/AbSciCon12
AbSciCon 2012 on Facebook : http://www.facebook.com/AbSciCon2012
News Contact
Marketing Communications Director
Parker H. Petit Institute for
Bioengineering & Bioscience
404-385-7001
Linda Billings
School of Media and Public Affairs
George Washington University
Chair, AbSciCon 2012
Communications Committee
Mar. 01, 2012
Robert M. Nerem, Ph.D., professor in mechanical engineering and Todd C.McDevitt, Ph.D., director of the Stem Cell Engineering Center at Georgia Tech,were invited by the lead sponsor, Semahat S.Demir Ph.D. of the National Science Foundation (NSF) to take part in aninternational assessment of the stem cell engineering field. Nerem willlead the panel and the findings of this study will result in recommendations tothe NSF and other funding agencies on future research directions andinvestments, recommendations on global initiatives with international partnersand public workshops.
The study, which is being conducted by the World Technology Evaluation Center(WTEC), aims to assess the current status and the trends of stem cellengineering, and compare U.S. research and development programs with thoseabroad. In addition to the NSF, the study is co-sponsored by the NationalInstitutes of Health (NIH) and the National Institute of Standards andTechnology (NIST).
“Tech is fortunate to have two out of the six experts on this panel,” Neremsaid. “It conveys Georgia Tech's nascent leadership in this relatively new andrapidly growing field and it is a great opportunity to provide input andleadership to our funding agencies and help our government understand wherebest to invest.”
President Obama, Congress and numerous states have recognized the value of stemcell research. Knowledge of research activities abroad will help to formulateand prioritize research directions to support President Obama's executive orderfor expanding stem cell research so that it has the greatest potential forclinical and commercial applications.
Dozens of companies have recently entered the stem cell engineering field insearch of clinical and commercial applications. There is clear impetusfor the U.S. to support stem cell research and continue its leadership in thebasic sciences for the betterment of humankind. A Congressional ResearchService report on stem cell research, which reviewed the political, moral andethical issues of the subject, indicated the strengthening interest andeconomic commitment for stem cell research in the U.S. and the rest of theworld.
This study will use WTEC's methodology and an expert panel of six to conductsite visits to overseas laboratories where work in stem cell engineering isdone. The panelists began their study in November, when they traveled to Chinaand Japan, and will continue their evaluation this week in Europe. Thesevisits, combined with the panel's own research experiences and assessments,will help shape a report. Like the previous WTEC studies on the tissueengineering and nanotechnology fields, this effort will act as a guide for U.S.research investments in this emerging field and will help identify key issuesof critical importance to program officers.
“This is an excellent opportunity to learn what other countries are doing andbenchmark against other programs in order to position the U.S. to becomeleaders in stem cell research and development,” said McDevitt, who is also anassociate professor in the Wallace H. Counter Department of BiomedicalEngineering at Georgia Tech and Emory University. “Manufacturing, clinicaltrials and commercializing stem cell-based products, if done strategically, issomething that could boost our nation’s economy.”
This week the scientists will travel to Denmark, France, Germany, Sweden and Switzerland. In addition to Nerem and McDevitt, other panelists include JeanneLoring, Ph.D., The Scripps Institute; Sean Palecek, Ph.D., University ofWisconsin; David Schaffer, Ph.D., University California at Berkeley; and PeterZandstra, Ph.D., University of Toronto.
WTEC is a non-profit 501(c)(3) research institute, which is a spin-off ofLoyola University Maryland. Since 1989, WTEC has provided such assessmentstudies in more than 60 fields of R&D under peer-reviewed grants from NSF.
News Contact
Marketing & Events
Parker H. Petit Institute for Bioengineering and Bioscience
Georgia Institute of Technology
Pagination
- Previous page
- Page 3
- Next page